Modelling of Water Turbidity Parameters in a Water Treatment Plant
A. S. KOVO
Chemical Engineering Department, Federal University of Technology, Minna, Nigeria
Abstract
The high cost of chemical analysis of water has necessitated various researches into finding alternative method of determining portable water quality. This paper is aimed at modelling the turbidity value as a water quality parameter. Mathematical models for turbidity removal were developed based on the relationships between water turbidity and other water criteria. Results showed that the turbidity of water is the cumulative effect of the individual parameters/factors affecting the system. A model equation for the evaluation and prediction of a clarifiers performance was developed:
Model: T = T0(-1.36729 + 0.037101∙10λpH + 0.048928t + 0.00741387∙alk)
The developed model will aid the predictive assessment of water treatment plant performance. The limitations of the models are as a result of insufficient variable considered during the conceptualization.
Keywords
Performance evaluation, pH, Alkalinity, Mathematic model, Simulation
1. Introduction
Tebbutt (1983) describes Water as the Worlds most important natural resource since without it life cannot exist and industry operate. Unlike many other resources, it has no substitute in many of its uses. Water plays an essential role in community development since a reliable supply is a prerequisite for establishing a permanent settlement.
There is a vast amount of water present in the earth and its surrounding atmosphere. About 7% of the earths mass is made up of water. 97% of this is found as saline water in oceans, about 2.3% is in the polar caps and only 0.7% exists in fresh water lakes, rivers, aquifers and in the atmosphere.
Water that is pure is not found in nature; even water vapour condensing in air contains solids and dissolved gases (Tebbutt, 1983). As it condenses and falls, it sweeps up other materials from the air, it becomes still more contaminated on reaching the ground, as it runs running over soil surface and percolate the soil strata.
Waste from human activities, either industrial or domestic, according to Tchobanoglous and Burton (1991), introduce even more pollutants, than any natural sources, into water bodies.
Since the available water is seldom found in conditions that meet potable specifications, a treatment plant is clearly necessary to improve the water quality. Water supply to a community goes through the following stages; the community water demand is carefully estimated with allowances for population growth; the most suitable raw water source is identified and analysed; then a water treatment plant to effect the required changes is designed, constructed and operated along with its distribution network.
When online, a periodic review of plant performance is undertaken to ascertain if, or otherwise, the plant works according to prediction. Hammer (1973) and Cairncross (1980) both agree that recordkeeping and periodic reviews of plant performance are necessary decision tools when the plant requires expansion or when operational problems arise.
The aim of this paper is to model water turbidity as function of several other parameters that will aid in assessing the performance of a water treatment plant over a period of time and to suggest ways of improving plant performance
2. Experiments
Determination of Temperature
The temperature of the sample of water was determined with the aid of thermometer, beakers, and electrodes. The water samples were collected in beakers and labelled appropriately to avoid mixing up. The thermometer is switched on and the electrode is dipped into the beaker in turn, the display is allowed to attain a steady value in each and the readings are recorded.
Determination of Turbidity
This involves the use of turbid meter. The sample cells were washing with sample water, and was discarded and refilled with the same sample. The sides of the cells were wiped with a dry cloth. The power source of the turbid meter was switch on and adjusted to read the turbidity in NTU. The cell holder was opened and the sample cell place in it and the lid replaced. The reading shows up on a digital display and the units are in NTU.
Determination of pH
The pH of the sample was determined with aid of colometric comparator. Two cells were rinsed with sample water and shaken, one cell was filled to mark B and 10 drops of indicator were added. The second cell was filled to mark A and placed in the comparator on the side marked blank. Slide the colorimeter through the slot in the comparator until the colour in both cells appears identical. The number in the matching colour slot is the pH.
Determination of Total Alkalinity
The total alkalinity was determined using titration. The burette was filled alkalimetric reagent up to the zero mark. 100ml of water sample was transferred to a breaker and add 4 to 6 drops of helianthin indicator was added. The Colour changes from pink to yellow. Titrate with the alkalimetric reagent until orange colour appears. Titrate with the alkalimetric reagent until orange colour appears. Read the alkalinity directly from the burette
3. Modelling for Turbidity Removal
For a constant flow rate and a fixed set of conditions the suspended solids (and hence turbidity) removal is a fixed fraction of inlet suspended solid. Assuming also, in accordance with Belan (1988), that for mildly contaminated rivers the suspended solid is linear with turbidity i.e. that the particle size and type distribution is constant over a time period.
Then Turbidity removal
(T0 T)/T0 = k0 (1)
where T = turbidity of clarified water, T0 = Turbidity of aerated water, k0 = constant
1 (T/T0) = k0, T/T0 = 1 - k0 = k', T = T0∙k' (2)
In reality operating conditions do vary. Therefore k' is only one of the function by which water quality may be measured. Other essential parameters according to Nikoladze et al (1989) are temperature (t), pH and alkalinity (alk). It is well established that these parameters exhibit independent and cumulative effect on T.
Effect of Water Temperature
Water molecules and impurity particles are in thermal Brownian motion whose intensity is directly proportional to temperature (Nikoladze et al, 1989). It is clear that the probability of collision of individual particles with one another and their consequent aggregation depend on their relative velocities i.e. on thermal Brownian motion (and therefore on water temperature).
Again, it shall be assumed that the temperature profile within the clarifier is constant and is equal to that of the clarified water samples.
(T0 - T)/T0 ~ t (3)
where t = temperature in °C.
Effect of Alkalinity
Turbidity removal is preceded by and dependent on the formation of chemical flock. The equation of the overall process as simplified by Hammer (1975) is
Al2(SO4)3 + 3Ca(HCO3)2 → 2Al(OH)3 + 3CaSO4 + 6CO2
Obviously, good coagulation is dependent on the presence of sufficient alkalinity (HCO3-) and therefore
(T0-T)/T0 ~ alk (4)
where alk = alkalinity of water in mg/l as CaCO3.
Effect of pH
Suspended matters in water are surfacecharged particles and it is the function of the coagulant to neutralize the charges. Different particles types have been seen to have a particular pH at which the net charge on them is zero and coagulation optimum (Nikoladze et al 1989). This pH is the isoelectric point (pHis). A large difference between pH of the water medium and pHis confers greater anticoagulation properties. For clay and humus pHis = 7.1 and 7.0 respectively. This effect is confirmed by graphs obtained by Tebbutt (1983) where turbidity (color) removal is reduced as the water pH deviates from an optimum value of about 7.0.
Therefore it can be concluded that:
(T0 - T)/T0 ~ 1/(pH - pHis) (5)
Assuming pHis as 7 then pH 7 = ∆pH has a positive value.
(T0 - T)/T0 ~ 1/(-∆pH) (6)
pH = - log[H+], therefore a more appropriate form of the equation would be:
(T0 - T)/T0 ~ 1/10-∆pH (7)
An expression similar to this was used by Belan (1988) to express the lime dosage required to being about a desired change in the pH of water to pHis of its contaminants.
Introducing constants into equations 3, 4 and 7:
(T0 - T)/T0 = k1/10-∆pH (8)
(T0 - T)/T0 = k2t (9)
(T0 - T)/T0 = k3alk (10)
Other factors, such as organic content do exert influence on turbidity removal; their contribution can be accounted for by the introduction of corrective coefficient.
Assuming that hydrodynamic conditions are approximately constant, the influences of temperature, pH, and alkalinity on the turbidity of water in a clarifier basin may be additive or multiplicative (Lucey, 2000). However a change in one will cause dis-equilibrium in the overall turbidity. Such changes are accounted for by the various constants. Using the additive method:
(T0 - T)/T0 = k0 + k1/10-∆pH + k2t + k3alk (11)
from which:
T = T0(1 - k0 + ki/10-∆pH + k2t + k3alk) (12)
Because 1 - k0 is a constant, let us say k0 in place of:
T = T0(k0 + k1/10-∆pH + k2t + k3alk) (13)
The values of these constants maybe determined using the least square method for multiple regression as outlined below adapted from Stroud (1995).
Let Ti be an observed value of outlet turbidity; the value predicted by the model equation is then:
TI = T0i(k0-(ki/10ΔpHi) - k2ti - k3alki) (14)
Then the error of prediction ei will be given by:
ei = Ti - T0i(k0-(k1/10ΔpHi) - k2ti - k3alki) (15)
The square of the error is:
ei2 = [Ti -T0i(k0 - (k1/10ΔpHi) - k2ti - k3alki)]2 (16)
For all data sets, by summing:
Σei2 = Σ [Ti - T0i(k0 - (k1/10ΔpHi) - k2ti - k3alki)]2 (17)
Expanding the brackets gives:
Σei2 = ΣTi2 + k02ΣT0i2 + k12ΣT0i2/(10ΔpHi)2 + k22ΣT0i2ti2 + k32Σt0i2alki2 -2k0k1ΣT0i2/(10δphi) - 2k0k2ΣT0i2ti - 2k0k3ΣT0i2alki + 2k1k2Σt0i2/(10ΔpHi) + 2k1k3ΣT0i2alki/(10ΔpHi) + 2k2k3ΣT0i2tialki 2k0ΣTiT0i + 2K1ΣTiT0i/(10ΔpHi) + 2k2ΣTiT0iti + 2k3ΣTiT0ialki
Partial differentiation of this equation with respect to the constants k0, k1, k2, and k3 gives:
∂(Σei2)/∂k0 = 2Σti.(∂Ti/∂k0) + 2k0ΣT0i2 - 2k1ΣT0i2/(10ΔpHi) -
- 2k2ΣT0i2ti - 2k3ΣT0i2alki - 2ΣTiT0i (18)
∂(Σei2)/∂k1 = 2ΣTi.(∂Ti/∂k1) + 2k1ΣT0i2/(10ΔpHi)2 - 2k0ΣT0i2/(10ΔpHi) +
+ 2k2ΣT0i2ti/(10ΔpHi) + 2k3ΣT0i2alki/(10δphi) + 2ΣTiT0i/(10ΔpHi) (19)
∂(σei2)/∂k2 = 2ΣTi.(∂Ti/∂k2) + 2k2ΣT0i2ti2 - 2k0ΣT0i2ti +
+ 2k1ΣT0i2ti /(10ΔpHi) + 2k3ΣT0i2tialki + 2ΣTiT0iti (20)
∂(σei2)/∂k3 = 2ΣTi.(∂Ti/∂k3) + 2k3ΣT0i2alki2 - 2k0ΣT0i2alki +
+2k1ΣT0i2alki/(10ΔpHi) + 2k2ΣT0i2tialki + 2ΣTiT0ialki (21)
Equating all the derivatives to zero yields the following system of equations
k0ΣT0i2 k1ΣT0i2/(10ΔpHi) - k2ΣT0i2ti k3ΣT0i2alki = ΣTiT0i (22)
k0ΣT0i2/(10ΔpHi) - k1ΣT0i2/(10ΔpHi)2 - k2ΣT0i2ti/(10ΔpHi) -
- k3ΣT0i2alki/(10ΔpHi) = ΣTiT0i/(10ΔpHi) (23)
k0ΣT0i2ti - k1ΣT0i2ti /(10ΔpHi) - k2ΣT0i2ti2 - k3ΣT0i2tialki = ΣTiT0iti (24)
k0ΣT0i2alki - k1ΣT0i2alki/(10ΔpHi) - k2ΣT0i2tialki - k3ΣT0i2alki2 =
= ΣTiT0ialki - (25)
The various effects of pH, temperature and alkalinity on turbidity removal may also be combined in a multiplicative or exponential manner since no evidence exists to suggest the relationship used. Following from the same premise as before, a possible second model equation such as 61 could be developed.
T = k0(T0∙10k1 ΔpHi∙tk2∙alkk3) (26)
Comparison of the models results indicates their extent of validity. The constants k0, k1, k2, k3 could be obtained from the solution of the generated 4´4 matrix.
The alternatively the developed models can be solved software packages such as Polymath3 (Himmelblau, 1996). The package was used to find the coefficients in both models. The developed models are presented below:
T = T0(-1.36729 + 0.037101∙10λpH + 0.048928∙t + 0.00741387∙alk) (Model 1)
T = 2.20673∙10-11∙T0·100.045606DpH·t5.35007·alk1.55059 (Model 2)
The graphical error analysis technique was then used as the criteria for selecting the best-fit model.
4. Results
The results of the various experimental methods are as presented in Table 1.
Table 1. Weekly water quality parameters values
|
Weeks |
Quality parameters |
||||
|
T0 |
T |
T |
DHp |
alk |
|
|
1 |
1.50 |
1.25 |
27.75 |
0.10 |
23.80 |
|
2 |
2.00 |
1.25 |
26.74 |
0.20 |
21.80 |
|
3 |
1.80 |
1.00 |
29.81 |
0.10 |
21.00 |
|
4 |
2.00 |
1.00 |
28.52 |
0.10 |
23.00 |
|
5 |
2.25 |
1.50 |
29.00 |
0.00 |
21.84 |
|
6 |
2.13 |
1.64 |
30.81 |
0.10 |
23.29 |
|
7 |
2.00 |
0.50 |
28.75 |
0.40 |
23.75 |
|
8 |
2.00 |
1.00 |
29.50 |
0.30 |
29.50 |
|
9 |
3.50 |
1.00 |
28.00 |
0.50 |
30.00 |
|
10 |
6.00 |
1.00 |
29.00 |
0.30 |
27.50 |
|
11 |
4.50 |
1.25 |
29.33 |
0.40 |
30.00 |
|
12 |
4.25 |
1.08 |
29.05 |
0.40 |
29.66 |
|
13 |
5.00 |
1.00 |
30.00 |
0.40 |
29.50 |
|
14 |
4.80 |
2.31 |
29.47 |
0.10 |
25.50 |
|
15 |
4.25 |
2.50 |
28.88 |
0.30 |
28.25 |
|
16 |
6.50 |
5.75 |
28.43 |
0.20 |
26.15 |
The simulated results from the models are presented in figure 1.
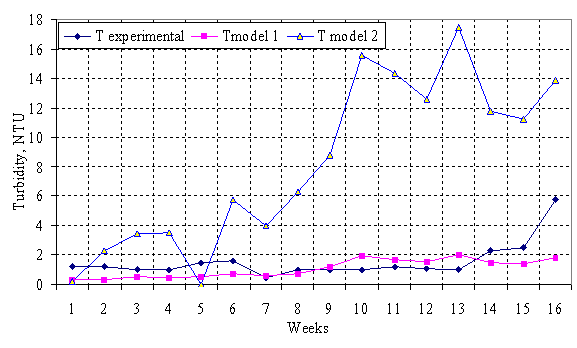
Fig.1. Comparative analyses of the experimental and simulated values from models 1 and 2
5. Discussions
Results from table 1 showed that turbidity in the treated water fell short of standards (5NTU max.) on one occasion (5.75NTU) but the mean value of 1.56NTU is well within the guide line value (WHO,1985). The cause of this is most likely the high T0 and the consequent clarifiers overload. This may also suggest that the clarifier was overdue for desludging.
Mathematical modeling of the clarifiers performance based on additive and multiplicative models using the pre- determined parameters gave models 1 and 2 respectively.
Model 1: T = T0(-1.36729+ 0.037101x 10λpH + 0.048928t + 0.00741387.alk)
Model 2: T = 2.20673 · 10-11To · 100.045606DpH · t5.35007 · alk1.55059
Simulation results of the models showed that model 1 to a large extent will give a better turbidity prediction. It showed also that factors affecting turbidity values are mainly independent in operation. The turbidity of water is the cumulative effect of the individual parameters/factors affecting the system.
From model 1 the change in turbidity caused by one of the independent variables is the cumulative effects of individual contributions of T0 , t, and alk. In model 2 the relationship of the turbidity parameters is as the synergy or interdependence of the variables. The nature of the model 1 equation gives turbidity change a dimensionless significance. Therefore k2 the coefficient of temperature t will have a unit of 1/oC. It could be defined as the change in ratio of inlet and outlet turbidity caused by changing the temperature of the clarifier by 1oC.
The discrepancies between the experimental and simulated values could be attributed to the assumptions made during the formulating of the model. The numbers of variables considered were quite small. The influence of human activities, impact of environmental pollution/contamination etc. on the coagulation and settling processes and much other need to be considered to enhance the reliability of the model.
Conclusions
Developed a model equation for the evaluation and prediction of a clarifiers performance as is presented below:
(Model 1) T = T0(-1.36729 + 0.037101∙10λpH + 0.048928∙t + 0.00741387∙alk)
The limitations of the models are as a result of insufficient variables considered during the conceptualization process.
Acknowledgement
We wish to acknowledge the contribution of the Federal University of Technology, Minna, Board of Research, for providing the necessary fund required for the execution of this research.
References
American Society of Civil Engineers/American Water Works Association (1990), Water Treatment Plant Design (2nd Ed.) Mc. Graw Hill, International, USA, 1990, p. 1-10.
Anderson R. (1987), Sample Pretreatment and Separation, John Wiley & Sons International, N.Y.C., 1987, p. 24-36.
Cairncross S. & Al (1981), Evaluation for Village Water Supply Planning, John Wiley & Sons Inc. N.Y.C., 1981, p. 2-15.
Fair G., Geyer J, Okun D. (1968), Water and Wastewater Engineering, (Vol.2), John Wiley & Sons Inc. N.Y.C., 1968, p. 27.1-27.12, 31.1- 31.29.
Hammer J.M. (1975), Water and Wastewater Technology, John Wiley & Sons, Inc, N.Y.C., 1975, p. 222-261.
Hellman H. (1981), Analysis of Surface Water, (English Ed.) Ellis Horwood Limited, 1981, p. 316-319.
Lucey T. (2000), Quantitative Techniques, 5th Ed., ELST, NY, 2000.
Midgley D., Torrance K. (1991), Potentiometric Water Analysis, (2nd Ed.), John Wiley & Sons International England, 1991, p. 55-89.
Nikoladze G., Mintis D., Kastalsky A. (1989), Water Treatment for Public and Industrial Supply, (English Ed.), MIR Publishers, Moscow, 1989, p. 51-70, 89-107, and 213-224.
Odigure J.O. (1998), Safety, Loss and Pollution Prevention in Chemical Process Industries, Jodigs & Associates, Nigeria, 1998, p. 18-22.
Owen H., Sandall O.C. (1983), Computational Methods in Chemical, 1983.
Tebbutt T.H.Y. (1983), Principles of Water Quality Control, 3rd Ed., Pergamon Press, England, 1983, p. 1-50, 125-140, and 168-174.
Tchobanoglous G., Burton F.L. (1991), Wastewater Engineering: Treatment, Disposal and Reuse, 3rd Ed., Mc Graw Hill, International, Calif., 1991, p. 104-108.
Twort A.C., Law F.M, Crowley F.W. (1985), Water Supply, 3rd Ed., Edward Arnold Publishers Ltd., Germany, 1985, p. 200-241.