Environmental Impact on the Quality of Water from Hand-Dug Wells in Yola Environs
Timothy Musa CHIROMA1, Benjamin Iyenagbe UGHEOKE2, David Onoja PATRICK1
1Chemical Engineering Department, Federal University of Technology, Yola, Nigeria
2Mechanical Engineering Department, Federal University of Technology, Yola, Nigeria
tchiroma@yahoo.com, ugheokeb@gmail.com, patondave@yahoo.co.uk
Abstract
The impact of environmental conditions on the quality of water from seven hand-dug wells in Vinikilang, Shinko, Demsawo and Girei was studied. Monthly physical and chemical analyses were carried out on the well water samples. The results revealed that the environment has direct impact on the quality of water and also the type of contamination of the well water samples. Water samples from the wells have higher levels of heavy metals: Fe, Zn, Cu and Pb, above the permissible limits of (0.1 mg/l, 5 mg/l, 0.5 mg/l and 0.05 mg/l for Fe, Zn, Cu and Pb respectively) WHO specifications, except well 1 whose Zn level was lower than the permissible limit. Wells close to abattoir, pit latrine, domestic refuse dumps, stagnant water and drainage showed higher amounts of coliform bacteria.
Keywords
Environment, Hand-dug wells, Water quality, Contamination, Health
Introduction
Yola is located in the North-East of Nigeria (longitude 09.40ºN and latitude 12.8ºE). It is characterized by two tropical climates marked by two seasons, namely, rainy and dry seasons, which run between May and October, and October to April, respectively. Vinikilang, Shinko, Demsawo, and Girei are towns within Yola Province, with characteristically rapidly growing populations. The development of unplanned houses to accommodate this rapidly growing population leads to the proliferation of refuse (waste) dumps, which invariably pose disposal problems. This is usually a common problem with many rapidly developing towns in Nigeria. The high population growth, poor development plan, chronic unhygienic habits and lack of enforcement of regulations have served collectively as recipe for environmental pollution. The problem of acute water supply has resulted in widespread use of hand-dug wells among which some are located in unhygienic areas. Studies in Japan (Shimizu et al, 1980) have shown that bacteria contaminate well water, depending on location. Thus, it is suspected that water from wells in these areas could be contaminated, according to their proximity to sources of pollutants.
The main objective of this study was to ascertain the quality of water samples from hand-dug wells in the selected towns in Yola environ, enumerated above. It also determined links between the environment of the wells and the nature of pollutants found in the well water.
Procedures
The well water samples were the main experimental materials. Seven wells were chosen for the study. This choice was borne out of their proximity to obvious sources of pollution, such as dumpsites, latrines and drainages. Special characters worthy of note among some of the hand - dug wells are summarized in Table 1.
Sample Collection
The well water samples were collected using 2 - liter hard plastic and screw-capped bottles that have been sterilized to avoid contamination by any physical, chemical or microbial means. The collected well water samples were aseptically transferred into sterile, 2- plastic containers. Samples for bacteriological analyses were kept in screw-capped bottles that have been sterilized in an autoclave for 15 minutes at 121ºC. Samples were then transferred to the laboratory where they were stored in the refrigerator for microbial analyses.
Table 1. Special Characteristics of the Wells
|
Well Sample No. |
Name of Town |
Nature and Dimension of Source |
|
1. |
Viniklang |
Well at 3m from drainage effluent |
|
2. |
Viniklang |
Well at from Abattoir |
|
3. |
Viniklang |
Well at 2m from irrigated farm and pool of stagnant |
|
4. |
Shinko |
Well in a residence house at 2m from pit latrine and 2m from refuse dump |
|
5. |
Shinko |
Well at 2m from domestic waste effluent |
|
6. |
Demsawo |
Well at 3m from dumpsite |
|
7. |
Girei |
Well at 3m from main road and drainage. |
Analysis of Well Water Samples
Sensitive parameters such as temperature and PH were measured immediately after collection of the well water samples. A Jenway 4015 pH meter and mercury thermometer were used for pH and temperature measurements, respectively. The conductivities of the well water samples were measured using a WPA cm - 35 electrical conductivity meter. To determine the total dissolved solids, about 100 ml of each sample was filtered, and the filtrate was measured in clean dried evaporating dish, which had its initial weight noted. The filtrate was evaporated to dryness on a hot water bath. The evaporating dish was cooled in desiccators for about 10 minutes and weighed with its content. The weighing was accomplished using a digital analytical balance (Ohaus, USA), with a sensitivity of 0.001g. The total dissolved solid of each sample was calculated using the formula:
Total dissolved solids = (A - B)/V*100
where A = Final weight of evaporating dish; B = Initial weight of evaporating dish; V = Volume of water sample taken (ml)
Trace and heavy metals in the well water samples were determined by Atomic Absorption spectrophotometer, pye Unicom SP 9 (AAS) using appropriate wave length for each metal (Fem Zn, Pb, and Cu). The turbidity of the samples was also determined using a turbidity meter. The bacteriological analysis for the presence of microbial and fecal contamination was investigated. The organisms were cultured for 24 hours using nutrient agar, a general-purpose agar for the culture of non-fastidious organisms and Mac Conkey agar, which is a selective medium for the isolation and differentiation of enteric organisms. The colonies were counted using Leica Quebec dark field colony counter.
Results and Discussions
The results of the analyses of the water samples from the seven hand-dug well in Yola environs, conducted from April to July 2005 are summarized in Tables 2 - 4.
Table 2. Variation of the Conductivity and Turbidity of the different Well Water Samples
|
Well No. |
Conductivity (us/cm) |
Turbidity (NTU) |
||||||
|
April |
May |
June |
July |
April |
May |
June |
July |
|
|
1 |
598 |
563 |
546 |
540 |
0.44 |
0.42 |
0.40 |
0.45 |
|
2 |
574 |
555 |
540 |
530 |
0.53 |
0.51 |
0.48 |
0.52 |
|
3 |
596 |
571 |
560 |
565 |
1.20 |
1.08 |
1.06 |
1.20 |
|
4 |
635 |
619 |
600 |
605 |
0.65 |
0.63 |
0.60 |
0.64 |
|
5 |
156 |
126 |
104 |
102 |
0.58 |
0.45 |
0.50 |
0.48 |
|
6 |
285 |
266 |
240 |
250 |
0.83 |
0.80 |
0.79 |
0.82 |
|
7 |
261 |
260 |
208 |
212 |
0.58 |
0.57 |
0.56 |
0.57 |
Table 3. Variation of the Total Dissolved Solids and salinity of the different well water samples
|
Well No. |
Total dissolved solids (mg/l) |
Salinity (mg/l) |
||||||
|
|
April |
May |
June |
July |
April |
May |
June |
July |
|
1 |
360 |
301 |
215 |
173 |
15.68 |
15.36 |
15.20 |
14.00 |
|
2 |
218 |
210 |
180 |
181 |
16.07 |
15.17 |
15.17 |
13.00 |
|
3 |
301 |
246 |
281 |
280 |
16.10 |
15.44 |
15.40 |
14.70 |
|
4 |
191 |
182 |
166 |
166 |
17.17 |
17.07 |
17.07 |
16.05 |
|
5 |
172 |
156 |
137 |
128 |
12.91 |
12.43 |
12.43 |
11.00 |
|
6 |
215 |
216 |
213 |
213 |
14.07 |
13.91 |
13.83 |
13.00 |
|
7 |
232 |
218 |
208 |
204 |
15.99 |
15.73 |
15.56 |
14.00 |
The pH of the well water samples was in the range of 6.5 to 8.5 as recommended by WHO (1971). The non-significant variations are consistent with the very little cleansing that takes place once the water has reached saturation (Domenico 1972). The mean temperature of the well water samples ranged from 29 to 30ºC which is about 2 to 3ºC cooler that the atmospheric temperature. The insulating property of the Earth dampens any extreme atmospheric condition on the well water samples. The turbidity values of the well water samples during the study period were generally lower than WHO maximum acceptable limit of 5.0 NTU.
Table 4. Variation of pH and bacteria levels of the different well water samples
|
Well No.
|
pH |
Coliform plate count |
||||||||||
|
Nutrient agar (Cfu/ 100ml) |
Mc Conkey agar (Cfu/ 100ml) |
|||||||||||
|
April |
May |
June |
July |
April |
May |
June |
July |
April |
May |
June |
July |
|
|
1 |
6.8 |
7.3 |
7.4 |
7.8 |
129 |
130 |
130 |
132 |
109 |
110 |
111 |
112 |
|
2 |
6.9 |
7.0 |
7.6 |
7.6 |
199 |
202 |
208 |
212 |
210 |
215 |
222 |
240 |
|
3 |
6.8 |
7.1 |
7.5 |
7.9 |
515 |
521 |
530 |
540 |
400 |
417 |
420 |
424 |
|
4 |
6.3 |
6.8 |
6.6 |
6.9 |
440 |
430 |
451 |
456 |
345 |
350 |
360 |
368 |
|
5 |
6.5 |
6.7 |
6.6 |
6.8 |
356 |
362 |
367 |
368 |
275 |
282 |
290 |
296 |
|
6 |
6.8 |
7.0 |
7.2 |
7.5 |
40 |
46 |
47 |
48 |
10 |
13 |
13 |
15 |
|
7 |
6.7 |
7.1 |
7.5 |
7.7 |
108 |
111 |
111 |
114 |
98 |
106 |
112 |
115 |
The conductivity of the well water samples ranges from 102 to 635 us/cm and the highest values of 530 to 635 us/cm were in wells 1, 2, 3 and 4 located in Viniklang and Shinko. The total dissolved solids of the well water samples were in general below the maximum acceptable limit of WHO (500mg/l). Although the salinity level of the well water samples was generally low (below the 200mg/1 maximum acceptable limit of WHO), well 4 had highest value, which fortunately is not inexplicable. Though, chlorine (Cl) is scarce in the soil, it is a common constituent of well water because of its mobility; hence it’s been used as a measure of pollution diffusion in ground water (Kaufman and Orlob, 1955). Septic tank leachates are known to pollute ground water with chloride (Sandhu et.al; 1978; Rajagol, 1978). It is thus not surprising that the well water sample with the highest salinity (17.17mg/l) was found in well 4, which is very close to pit latrine.
The levels of heavy metals in well water samples are generally higher than the maximum acceptable limit of WHO. Fe level in all the wells was above the WHO maximum acceptable limit of 0.1mg/1. Higher level of 0.37 and 0.40mg/1 is from wells 6 and 3, respectively, situated near dumpsite, irrigated farms and sewage waters. Thus the high level of Fe in this well water samples has been attributed to their locations near such source, a trend already observed by researchers (Sonumacher et al, 1993). Significant level of Zn was detected in the well water samples from wells 3,4,5,6 and 7. Level as high as 28.84 mg/1, which is 5.8 times the WHO maximum allowable limit of 5 mg/l for Zn, was detected in water sample of well 6. These wells are located close to dumpsite, domestic wastes and drainages where all sorts of metal wastes are found. The leaching of these metals by rainfall is the probable cause of the high levels of these pollutants in the well water samples of these wells. The level of Cu in all the water samples was more than 10 times above the WHO maximum limit of 0.5 mg/l. According to UNESCO and WHO (1984), copper is rarely found in natural water, consequently, its existence in high concentration in the well water within the area of study is an index of pollution from leachates and run effluents of the polluted environments where the wells are located. The Pb level ranged from 4.3 mg/l to 12.78 mg/l which is about 86 to 256 times the maximum allowable limit of 0.05 mg/l specified by WHO. Well 7 which is located closer to the main road and drainages has the highest value of lead, which is not a surprise since automobile emissions is one of the sources of pollution of the environment (Iserman, 1977 and Essien, 1996). The consistently high value of Pb level in the well water samples is quite disturbing since Pb is a well known poison and the well water is a source of portable water for most of the communities in the area, thus the danger of Pb poisoning becomes very real for the users. The variation of the concentration of the metals Fe, Zn, Cu, and Pb in the different wells under study are shown in Figures 1 to 4.
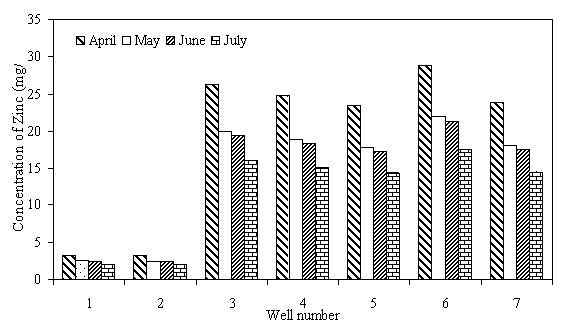
Figure 1. Zinc (Zn) levels in well water samples
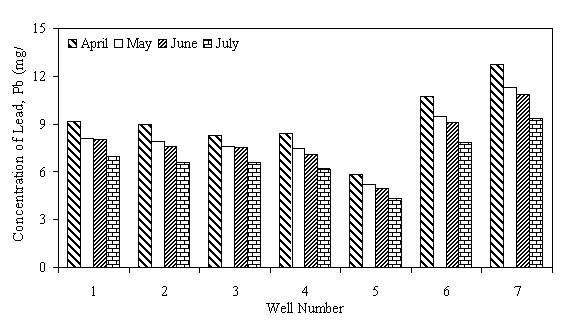
Figure 2. Lead (Pb) levels in well water samples
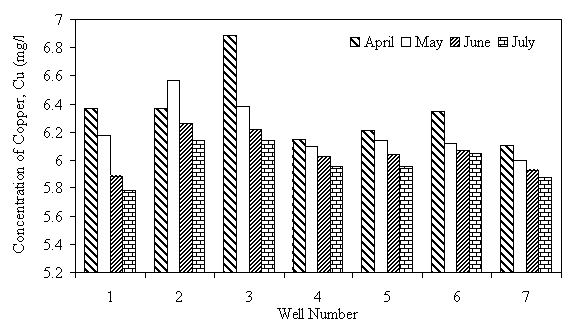
Figure 3. Copper (Cu) levels in well water samples
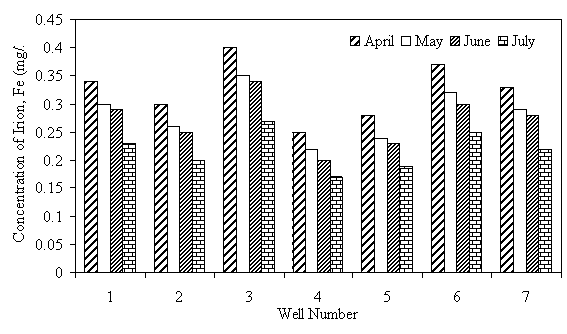
Figure 4. Iron (Fe) levels in well water samples
Bacteriological analysis of all the water samples except well 6 (located 3m from dumpsite) shows a higher number of Coliform bacteria. These wells are located either too close to domestic refuse waste, abattoir, pit latrine, stagnant water or drainages. It is possible that in spite of the too many to count coliform bacteria, Escherichia coli may still be absent (Shimizu et al, 1980 and Schuber and Tinki, 1998). However, it is still a source of concern because it can be a source of many diseases to the unwary or ignorant. It could not be ascertained whether the wells were dug unwisely close to these sources of pollutants or the sources of pollutants were created after the wells had been dug. Baring any act of desperation or share ignorance, the later is most likely to be the case. Facilities meant for a few are very often over capacitated because they cannot cope with the rapid growth in population or changing conditions. Water supply and waste disposal are no exceptions to general problems in developing countries like Nigeria.
These results are consistent with the findings of Shimizu et al (1980) whose studies of over 318 wells in Japan showed more microbial activities were found in wells close to organic waste sites.
Conclusion
The results revealed that the qualities of the water samples were affected by the conditions of the immediate environment. Wells located either close to domestic refuse dumps, abattoir, pit latrines, stagnant water or drainages showed high value of coliform bacteria.
Generally, all the wells located close to sources of heavy metal pollutants have the values of Fe, Cu, Zn and Pb above the WHO maximum permissible value except wells 1 and 2 that had its Zn value below the WHO maximum value.
From the above conclusion, it follows that the populaces need to be educated about the dangers of using such well waters without treatment.
More so, there is a need to create awareness about the influence of environment on the quality of water obtained from wells. This should of course, go a long way to forestall any looming danger from water borne diseases and water pollution.
References
[1] Domenico P. A., Concepts and Models in ground water hydrology, New York, McGraw Hill pp.279-314, 1972.
[2] Essien I. O., Distribution of lead in Akwa Ibom State, Global J. Pure Appl. Sci. 2 (1), 1996, p. 29-35.
[3] Iserman K. A., A method to reduce the contamination and uptake of lead by plants from car exhaust gases, Journal of Environmental Pollution, 12, 1977, p. 199-203.
[4] Kanfman W. J., Orlob G. T., Measuring ground water movement with radioactive chemical tracers, Journal of American water works Association, 48, 1955, p. 559-572.
[5] Rajgopal R., Impact of land use on ground water quality in the grand transverse bay of Michigan, Journal of Environmental Pollution, 7, 1978, p. 93-98.
[6] Sandhu S. S., Waren W. J., Nelson S. P., Trace inorganic substances in rural portable water and their correlation to possible sources, Water Research, 12, 1978, p. 257-261.
[7] Schuber B. J., Tinka M. A., Microbiology of sewage effluents, Water Engineering, 1989, p. 451- 456.
[8] Shimizu T. T. M., Kigotake T. M., Agatomo H. N., Bacteria contamination of drinking water from wells in Miyazaki, Japan, Bull Foe. Agric. Miyakazi Univ., 1980, p. 21-28.
[9] Sonuhmacher M., Domingo J. L., Liobet J. M., Conbella I. J., Chromium, copper and zinc concentration in edible vegetables grown in Tarranga Province, Spain, J. Environmental Contamination and Toxicology, 58, 1993, p. 515-521.