Phytoremediation of Heavy Metals in Aqueous Solutions
Felix Aibuedefe AISIEN1,*, Oluwole FALEYE1 and Eki Tina AISIEN2
1Department of Chemical Engineering, University of Benin, Benin City, Nigeria/
2Department of Integrated Science, College of Education Ekiadolor-Benin, Nigeria/
E-mails: afaisien2002@yahoo.com, ekiaisien@yahoo.com
* Corresponding author
Received: 26 September 2010 / Accepted: 21 December 2010 / Published: 30 December 2010
Abstract
One of the major environmental problems is the pollution of water and soil by toxic heavy metals. This study investigated the phytoremediation potential of water hyacinth, for the removal of cadmium (Cd), lead (Pb) and zinc (Zn). Water hyacinths were cultured in bore-hole water, supplemented with 5mg/l of Zn and Pb and 1mg/l of Cd at pH 4.5, 6.8 and 8.5. The plants were separately harvested each week for six weeks. The results showed that removal of these metals from solution was fast especially in the first two weeks, after which it became gradual till saturation point was reached. The accumulation of Cd and Zn in leaves and roots increased with increase in pH. The highest accumulation was in the roots with metal concentration of 4870mg/kg, 4150mg/kg and 710mg/kg for Zn, Pb and Cd respectively at pH 8.5. The maximum values of bioconcentration factor (BCF) for Zn, Pb and Cd were 1674, 1531 and 1479 respectively, suggesting that water hyacinth was good accumulator of Zn, Pb and Cd, and could be used to treat industrial wastewater contaminated with heavy metals such as Zn, Pb and Cd.
Keywords
Phytoremediation; Heavy metals; Water hyacinth; Bioaccumulation; Leaves; Roots
Introduction
The removal of heavy metals constituents from industrial wastewater is of paramount important taken into consideration the environmental problems associated with soil and water polluted with heavy metals. Tyler et al, [1] reported that the release of heavy metals in biologically available forms by human activity may damage or alter both natural and man-made ecosystems. One of the effective and affordable technological solutions is the application of a process known as phytoremediation. Phytoremediation is the application of plants for in situ or ex situ treatment/removal of contaminated soils, sediments and water. The green plants degrade, assimilate, metabolize, or detoxify inorganic and organic pollutants from the environment or render them harmless. It is a cost effective green technology based on the use of specially selected metal-accumulating plants to remove toxic metals from soils and water [2]. Apart from the advantage of low cost, extraction and concentration of a particular element from the environment, the harvested plants tissue, rich in accumulated contaminants, is easily and safely processed by drying, ashing or composing. The metals can then be reclaimed from the ash, which further reduces the generation of hazardous waste and generate recycling revenue. However, despite these advantages, phytoremediation has not been widely used in developing country such as Nigeria. This is because of the limited knowledge of this technology. Due to inappropriate legislation, presently in Nigeria industrial waste that contains mainly toxic heavy metals are discharged without treatment into the environment thereby contaminating soils, underground water and surface water. The aftermount effects include:
§ Destruction of eco system;
§ Destruction of marine biota;
§ Hindering of fisheries and other aquacultural operations;
§ Poisoning of sea food, contamination of surface and underground water;
§ Disturbances of soil function;
§ Reduction of plants productivity and chance of survival etc.
However, some elements such as Cu2+, Zn2+, Fe2+, are essential micronutrients for plant metabolism but when present in excess, can become extremely toxic. Cadmium (Cd) is one of the most toxic heavy metals and is considered non-essential for living organisms. Zinc (Zn) is an essential and beneficial element for human bodies and plants. Lead (Pb), on its own has been discovered to be non-toxic to plants [3].
Water contaminated with heavy metals has been treated in the past by the use of ion exchange and activated charcoal filter methods. The disadvantages of this processes is that they are expensive and nonselective for heavy metal removal. As a result, phytoremediation, a low cost and effective and efficient method is now being used. Microorganisms and terrestrial plants have been used in recent time [4]. However, for the purpose of this study, aquatic plant, water hyacinth (Eichhornia crassipes) was employed as a plant model for the removal of heavy metals from aqueous solution. Water hyacinth plant causes severe environmental and economic problems in the Niger-Delta area in Nigeria especially along the coastal water-way. Soltan and Rasheed, [5] also reported that water hyacinth is one of the most productive plants on earth and is consider as the worlds worst aquatic plant. It forms dense mats that interfere with navigation, recreation, irrigation, and power generation. It also pollute surface water making it unfit for consumption. The plant accumulates toxic metals such as Cd, Pb and Zn, directly from water, which serves as sources of nutrients.
Consequently, the aim of this study was to use water hyacinth plant in the:
§ Determination of the potential of water hyacinth in accumulating heavy metals (BCF).
§ Determination of the effect of pH on metals adsorption in aqueous solution.
§ Determination of which of the plant tissue (roots or leaves) adsorbs more heavy metals.
Material and Method
Material Collection
The water hyacinth plant (E. crassipes) was collected from a pond in Botany Department, University of Benin, Benin-City, Nigeria. Analytical grade PbCl2. 5H2O, CdCl2. 5/2H2O and ZnSO4. 7H2O were obtained from Chemical Engineering laboratory in University of Benin, Benin City, Nigeria.
Samples Preparation
Stock solutions 1mg/l of Cd, 5mg/l Zn and 5mg/l Pb each for pH 4.5, 6.8 and 8.0 were prepared in distilled water with analytical grade PbCl2. 5H2O, CdCl2. 5/2H2O and ZnSO4. 7H2O. Ten (10) medium size plants were placed in each 40 litre plastic tank containing bore-hole water supplemented with the stock solutions and placed under natural sunlight. Plants in only bole-hole water tank served as control. The test duration was for six (6) weeks. At the end of each week, plants were harvested. They were separated into shoots and roots, and were analyzed for metals accumulation, and the bioconcentration factor (BCF) was determined. Also water samples were collected for metals analysis each week.
Metals Analysis
Metals accumulated in plant tissues and water samples were measured. The plants tissues (leaves and roots) were digestion using the standard methods by APHA, [6]. Plant samples were decomposed to dry matter by heating at 120°C for 24 hours in a hot air oven and the ash was digested with nitric acid and filtered into a volumetric flask. The final volume was made up with deionized water and heavy metals analysis was done using a Solar 969 Unicam series atomic absorption spectrophotometer (AAS). The concentration of metals that remains in the residual solution was also measured using AAS. The difference between the initial metal concentration and remaining metal concentration in the solution was taken to be metals bound to the plant.
The ability of the plant to accumulate metals with respect to the metal concentration in the substrate is known as the bioconcentration factor (BCF). Zayed et al, [7] reported that BCF can be calculated as follows:
BCF = Concentration of metal in plant tissue / Initial Concentration of metal in external solution.
Results and Discussion
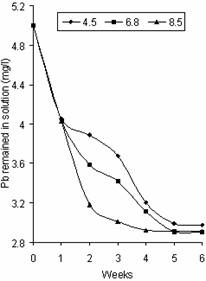
Figure 1 shows the variation of Pb concentration in solution with pH. The results at different pH values show an initial rapid decrease of Pb in the solution within the first two weeks from 5mg/l to 3.89mg/l, 3.59mg/l and 3.18mg/l for pH values 4.5, 6.8, and 8.5 respectively. However, a slight decrease was observed in the third week to 3.01mg/l for pH 8.5, after which the value remained relatively constant at an approximate value of 2.91mg/l. Concentration of Pb for pH values 4.5 and 6.8 in the solution continued to decline at a slower rate from week 2 to week 5 after which it remained constant at 2.98mg/l and 2.90mg/l respectively. It is apparent that as pH increased, adsorption increased and the concentration of Pb in solution decreased. These results agreed with that reported by Lopez et al, [8].
|
Figure 1. Variation of Pb concentration in solution with pH |
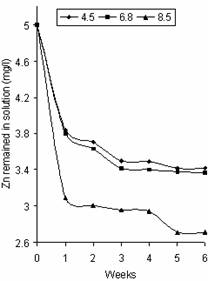
Figure 2. Variation of Zn concentration in solution with pH |
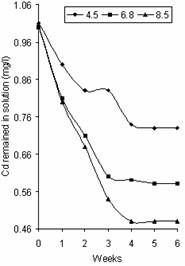
Figures 2 and 3 represent the variation of Zn and Cd concentration in solution with pH respectively. The profile of these figures is similar to that of Figure 1 except that most of the Zn was adsorbed in the first week at different pH values, from an initial concentration of 5mg/l to 3.09mg/l, 3.80mg/l and 3.84mg/l at pH 8.5, 6.8 and 4.5 respectively. A slight decrease was observed in week 2 at pH 8.5 and within week 2 and week 3 at pH 6.8 and 4.5 after which adsorption became relatively stable reaching the saturation points of 3.42mg/l, 3.37mg/l and 2.71mg/l at pH 4.5, 6.8 and 8.5 respectively. Cd adsorption decreased gradually through the first four weeks for pH 8.5, the first three weeks for pH 6.8 and the first two weeks for pH 4.5 with values 0.48mg/l, 0.60mg/l and 0.83mg/l respectively. The saturation points for Cd were 0.73mg/l, 0.58mg/l and 0.48mg/l at pH values 4.5, 6.8 and 8.5 respectively. The trend of increase intake of cationic metals with increase pH was observed in Figures 2 and 3. This agrees with that reported by Cheng and Allen [9].
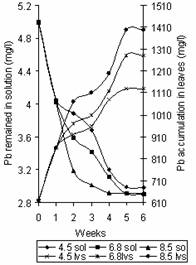
Figure 4 shows the variation of Pb concentration in solution and accumulation in leaves with time. The observed trend correlates with that in Figure 1. This was explained by Lopez, et al. [8] that as pH increases, the binding of metal to the plant tissues also increases. It was also shown in Figure 1 that the binding of most of the Pb present in solution to the plant tissue occurred within the first two weeks of planting. This statement can be justified with the profile shown in Figure 4. It can be seen that the concentration of Pb in the plant tissue increased from an initial value of 620mg/kg to 1070mg/kg, 970mg/kg, and 920mg/kg at pH 8.5, 6.8 and 4.5 respectively. The saturation point in the leaves was reached at the end of week 5 with Pb concentration of 1400mg/kg, 1280mg/kg and 1130mg/kg at pH values of 8.5, 6.8 and 4.5 respectively. The corresponding values of Pb concentration in solution were 2.91mg/l, 2.90mg/l and 2.98mg/l respectively.
|
Figure 3. Variation of Cd concentration in solution with pH |
Figure 4. Variation of Pb concentration in solution and time accumulation in leaves |
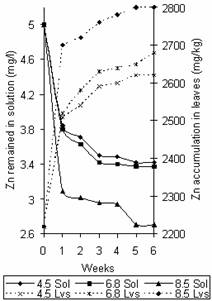
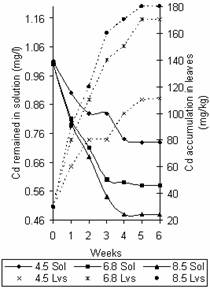
Figures 5 and 6 show the variation of Zn and Cd concentration in solution and that accumulated in leaves with time. The observed trend agrees with that of Figures 2 and 3 for Zn and Cd respectively. Also it was observed that most of the Zn binding occurred in the first week while that of Cd increased gradually through the weeks at the different pH values. The metal concentration in leaves at saturation points recorded were 2800mg/kg, 2680mg/kg and 2620mg/kg for Zn and 180mg/kg, 170mg/kg and 111mg/kg for Cd at pH values 8.5, 6.8 and 4.5 respectively.
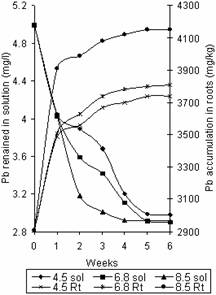
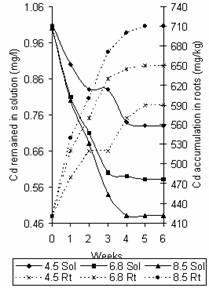
Figure 7 represents the variation of Pb concentration in solution and that accumulated in roots with time. The same trend of increased intake at increased pH was also observed. However, differences lies on the point of saturation with Pb concentration of 4150mg/kg, 3810mg/kg and 3740mg/kg at pH 8.5, 6.8 and 4.5 respectively. The corresponding values of Pb concentration in solution were 2.91mg/l, 2.90mg/l and 2.98mg/l respectively.
|
Figure 5. Variation of Zn concentration in solution and time accumulation in leaves
|
Figure 6. Variation of Cd concentration in solution and time accumulation in leaves
|
|
Figure 7. Variation of Pb concentration in solution with pH |
Figure 8. Variation of Zn concentration in solution and time accumulation in leaves |
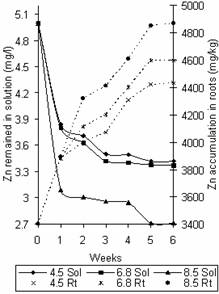
Figures 8 and 9 show the variation of Zn and Cd concentration in solution and that accumulated in roots with time. The trend observed in these figures agrees with that reported by Zaranyika and Ndapwadza [10]. The saturation points for the accumulation of Zn in the roots were 4870mg/kg, 4600mg/kg and 4430mg/kg, while that for Cd were 710mg/kg, 650mg/kg and 590mg/kg at pH values 8.5, 6.8 and 4.5 respectively.
|
Figure 9. Variation of Cd concentration in solution and time accumulation in roots |
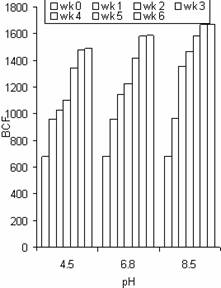
Figure 10. The bioconcentration factor (BCF) values of Zn in water hyacinth at different pH and exposure time |
This research studies show that water hyacinth accumulated the highest concentration of metals in roots at pH 8.5, with values 4150mg/kg, 4870mg/kg and 710mg/kg for Pb, Zn and Cd respectively. However, relatively little was translocated to the leaves with concentration 1400mg/kg, 2800mg/kg and 180mg/kg for Pb, Zn and Cd respectively. This trend of higher accumulation in roots than in leaves agrees with that reported by Stratford et al. [11].
|
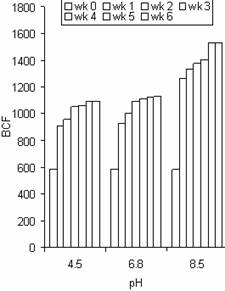
Figure 11. The bioconcentration factor (BCF) values of Pb in water hyacinth at different pH and exposure time |
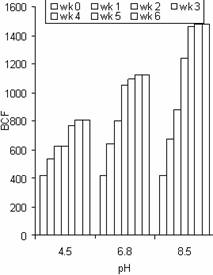
Figure 12. The bioconcentration factor (BCF) values of Cd in water hyacinth at different pH and exposure time |
Zhu et al., [12] also reported that the main route of heavy metal uptake in wetland plants was through the roots in case of emergent and surface-floating plants like water hyacinth and that much of the accumulation into the plant tissue is by absorption to the anionic sites in the cell walls. This explains why wetland plants can have very high magnitude of heavy metal concentration in their tissues compared to their surrounding environment.
The BCF values of Zn at different pH in the roots were a little higher than that of Pb and Cd for the same duration in most cases. The maximum BCF values for Zn, Pb, and Cd were 1674, 1531 and 1479 respectively. These results indicate the accumulation potential of Zn by water hyacinth was slightly higher than that of Pb and Cd. The results also confirm the use of water hyacinth as a good hyper accumulator of Zn, Pb and Cd based on the arbitrary criteria given by Zayed et al., [7] reported that the appropriateness of a plant for phytoremediation potential is often judged by its BCF. BCF values over 1000 are generally considered evidence of a useful plant for phytoremediation. This study gave BCF values above 1000 for each metal, indicating water hyacinth plant is a good hyperaccumulator of Zn, Pb and Cd.
Conclusions
The following conclusions can be drawn from this study:
§ Zn, Pb and Cd bind to water hyacinth roots and leaves and the binding was observed to be pH dependent, with the highest binding occurring at a higher pH of 8.5.
§ There was more bioaccumulation of metals in the roots of water hyacinth than to the leaves.
§ The accumulation potential of Zn by water hyacinth was higher than that of Pb and Cd.
§ Water hyacinth may be used in environment technology in constructed wetlands as it has been shown to be a good hyperaccumulator of Zn, Pb and Cd with BCF values greater than 1000.
References
1. Tyler G., Pahlsson A. M., Bengtsson G., Baath E., Tranvik L., Heavy metal ecology and terrestrial plants, microorganisms and invertebrates: a revie,w Water Air Soil Pollution, 1989, 47(3-4), p. 189-215.
2. Garbisu C., Alkorta I., Phytoextraction: A cost effective plant-based Technology for the removal of metals from the environment, Bioresource Technol., 2001, 77(3), p. 229-236.
3. Gupta G. C. Use of water hyacinth in wastewater treatment (a brief literature review), J Environ. Health, 1980, 43(2), p. 80-82.
4. Sadowsky M.J. Phytoremediation: past promises and future practices. In: Proceedings of the 8th International Symposium on Microbial Ecology (Edited by Bell C.R., Brylinsky M. and Johnson-Green P.). Atlantic Canada Society for Microbial Ecology, Halifax, Canada, 1999.
5. Soltan M. E., Rasheed M. N., Laboratory study on the survival of water hyacinth under several conditions of heavy metal concentrations, Adv. Environ. Res., 2003, 7(2), p. 321-334.
6. APHA, AWW, WEF, Standard Methods for the Examination of Water and Wastewater. 20th Ed., Washington DC, 1998.
7. Zayed A., Gowthaman S., Terry N., Phytoaccumulation of trace elements by wetland plants: I. Duckweed, Environ. Qual., 1998, 27(3), p. 715-721.
8. Lopez A., Lazaro N., Morales S., Marques A.M. Nickel Biosorption by free and immobilized cells of Pseudomonas flourescens: A Comparative Study, Water, Air, Soil pollution, 2002, 135(1-4), p. 157-172.
9. Cheng T., Allen H.E., Prediction of Uptake of Copper from solution by Lettuce. Environ. Toxicol. Chem., 2001, 20(11), p. 2544-2551.
10. Zaranyika M.F., Ndapwadza T., Uptake of Ni, Zn, Fe, Co, Cr, Pb, Cu and Cd by water hyacinth (eichhornia crassipes) in mukuvisi and manyame rivers, Zimbabwe, Journal of Environmental Science and Health, 1995, 30(1), p. 157-169.
11. Kay S.H., Haller W.T., Garrard L.A., Effects of heavy metals on water hyacinths, Aquat. Toxicol., 1984, 5(2), p. 117-128.
12. Zhu Y.l., Zayed A.M., Qian J.H., Souza M., Terry N., Phytoremediation of trace elements by wetland plants: II. Water hyacinth, J. Environ. Qual., 1999, 28, p. 339-344.