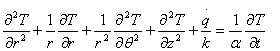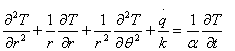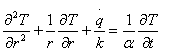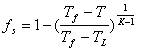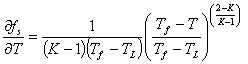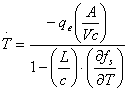Rate of solidification of aluminium casting in varying wall thickness of cylindrical metallic moulds
Katsina Christopher BALA*, Reyazul Haque KHAN
Department of Mechanical Engineering, Federal University of Technology, P.M.B. 65, Minna, Nigeria.
E-mails: chrisbalamaik@yahoo.co.uk; reyazkhan1@yahoo.com
* Corresponding author, Phone: +2348035980302
Abstract
The quality of final casting mainly depends on the rate of solidification as rapid solidification produces fine grains structures with better mechanical properties. The analysis of heat transfer during the casting and solidification of aluminium alloy as well as the experimental investigation of the rate of solidification in varying thicknesses of cylindrical metallic mould was carried out. The temperature variation with time of the casting was recorded from which cooling curves were obtained for the determination of solidification time of the cast. The results showed that as the cylindrical mould thickness increases the solidification time decreases due to the chilling effect of the mould.
Keywords
Casting; Solidification; Metallic mould; Aluminium alloy; Heat transfer; Cylindrical mould; Temperature; Heat conduction; Solidification time
Introduction
Metal casting is a process of pouring molten metal into the cavity of a mould and allowed to solidify into a required shape. This is an ancient method [1] used to manufacture complex parts of components and equipment by imparting desirable characteristics of the casting for specific area of application as in transportation [2], communication [3], power [3], agriculture [4], construction [5], Airspace [2], chemical, petrochemical[4] and other industries. However the quality of the final casting is dependent upon many parameters such as: pouring temperature, initial temperature of the mould, shape and size of the mould, time of pouring into the mould, and composition of the metal [6]. Similarly, the solidification time of the casting is dependent upon the various parameters like mould wall temperature, heat transfer coefficient at the metal-mould interface, mould wall thickness, material of the mould [7].
The mould is a cavity where molten metal is poured to solidify and form the desired shape. Among the parameters listed above, the mould is one of the major parameter that determines the outcome of other parameters. The rate at which a casting solidifies will affects its microstructure, quality, and mechanical properties. Generally speaking, an area of the casting which is cooled quickly will have a fine grain structure and an area which cools slowly will have a coarse grain structure [8].
Metallic (permanent) mould as the name implies are made of metal capable of withstanding high temperature, coated with refractory wash to reduce its chilling effect on the cast and facilitates the removal of cast product. In practice it is frequently necessary to know how the rate of solidification of a casting changes depending upon the configuration, material and weight of the mould. Bala et al. [9] investigated the rate of solidification in permanent rectangular moulds of varying thicknesses.
Solidification of metals is the phase transformation of molten metal into solid state after pouring into the mould [10]. This process begins immediately the molten metal is poured into a mould and it is accompanied by series of events. The essential feature of solidification is the liquid to solid phase transformation along a moving interface, which is accompanied by the release of latent thermal energy and its transfer through both phases. This change of phase of the material differs, depending on whether the material is a pure element or an alloy [11]. A pure metal solidifies at a constant temperature, which is its melting point (freezing point). For alloys, the solidification occurs over a temperature range depending upon the composition. The cooling characteristic which brings about solidification have a considerable influence on the grain size [12] and the microstructure of the cast part, there by determining the mechanical properties as well as some metallurgical factors [6].
Solidification of metal casting is dependent on the rate of heat removal from the metal to the mould. This determines the solidification time, temperature, and microstructure. Therefore the understanding of the effect of mould thickness, time, and temperature can be a useful tool with which to improve process design and in turn control product quality. So this study assessed the effect of mould thickness on solidification time of a cylindrical cast to help improve casting process thereby producing high quality products.
Material and method
The development of a mathematical model for heat transfer during solidification in cylindrical coordinate systems is presented. This heat transfer is governed by the principles of classical thermodynamics for the conservation of energy. The energy equation along with the equation of state and boundary conditions determine the distribution of temperature in the molten metal as well as the metallic mould.
The materials used for the experimental investigation of the solidification process were three cylindrical gray cast iron moulds of varying thicknesses of 12.5mm, 37.5mm and 50.0mm; aluminium alloy (LM14) of composition: Cu 4%, Mg 1.5%,Ni 2% and Al 92.5% obtained from scrap pistons; resistance furnace for melting; paperless temperature recorder; K-type thermocouples as temperature sensors.
Heat transfer during solidification
Two important phenomena in solidification are: phase change and heat transfer. In an adiabatic system (i.e. no heat transfer), solidification cannot take place. Heat transfer can occur by conduction, convection and radiation [13]. In conduction, the driving force is the temperature gradient. Heat transfer rate per unit area is proportional to the normal temperature gradient given by Fourier law as
|
|
(1) |
where q =
heat transfer rate, A = surface area, k = thermal conductivity of
the material, ![]() temperature
gradient in the direction of heat flow.
temperature
gradient in the direction of heat flow.
In convection heat transfer, the driving force is the motion of fluid over the surface. By Newton’s law of cooling,
|
|
(2) |
where Tw = temperature of surface, T∞ = temperature of fluid, h = convection heat-transfer coefficient
Unlike conduction and convection, no medium is required for heat transfer in radiation. It is the fastest way of heat transfer since it takes place through electromagnetic radiations which travels at the speed of light. According to Stefan-Boltzmann Law an ideal thermal radiator (i.e. a perfect blackbody) emits energy thus:
|
|
(3) |
where σ = Stefan-Boltzmann constant (5.669×10-8W/m2K4)
Heat conduction in cylindrical coordinates
The mathematical model for conduction in cylindrical shape is based on the general equation of heat conduction, expressed in cylindrical coordinates (x, θ, z) system [14] is given by:
|
|
(4) |
where r, θ and z are the cylindrical coordinates represented in Figure 1.
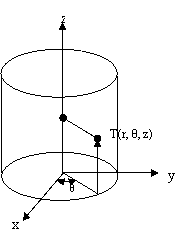
Figure 1. Cylindrical coordinate system
Considering that the flow of heat is mostly radial, equation (4) can be reduced to a two-dimensional form. Thus in the z-direction heat extraction can be neglected, since it is not significant as compared to the heat flow in r and θ directions [15] to yield:
|
|
(5) |
For ingots with a symmetrical section, heat flow in θ direction can also be neglected. Hence,
|
|
(6) |
But heat liberated during solidification (q) in the unsteady state condition equation (6) for cylindrical shape is expressed as:
|
|
(7) |
where L = latent heat of fusion, fs = solid fraction, t = time.
Relating equation (7) to temperature thus:
|
|
(8) |
Substituting equations (7) and (8) into equation (6) we obtain
|
|
(9) |
where
|
|
(10) |
If the liquid is uniform in composition throughout solidification, and has a composition of CL, then the solid phase at the interface is given by:
|
|
(11) |
where K = partition coefficient.
The fraction of molten metal solidified is,
|
|
(12) |
where Tf = fusing temperature of pure substance, TL = liquids temperature.
Equation (12) is Scheil equation [15], which estimates for aluminium alloy the fraction of solid in the mushy zone. In obtaining the equation it was assumed that perfect mixing in the liquid and no solid diffusion, i.e. the diffusion of alloying elements in the solid phase is slow.
Differentiating equation (12) with respect to temperature by chain rule we have
|
|
(13) |
In solidification process, extraction of heat is achieved by applying a suitable means of cooling to the melt in order to create an external heat flux, qe. The resultant cooling rate can be deduced from a simple heat balance if the metal is isothermal (low cooling rate) and the specific heats of the liquid and the solid are the same. Using the latent heat per unit volume, L, (defined to be positive for solidification), and also the specific heat per unit volume, c, then the cooling rate is expressed [15] as:
|
|
(14) |
where A =surface area of casting, V = volume of cast.
Experimental method
To investigate the influence of mould thickness on the solidification time of aluminium alloy cast in cylindrical moulds experiments were performed to obtain solidification time and the temperature profiles within the aluminium cast and the cylindrical moulds. The moulds thicknesses were varied while the cast (mould cavity) volume remained constant. Three different mould thicknesses of 12.5 mm; 37.5 mm and 50.0 mm with a mould cavity of depth of 100 mm and diameter of 100 mm were used. Table 3.3 gives the various dimensions of the cylindrical moulds. The moulds were assembled together using brackets, nuts and bolts.
Aluminium alloy obtained from scraps of pistons were charged in a metallic crucible and placed in a resistance furnace until it melted and attained a temperature of 680°C.The molten aluminium metal was then taken out of the resistance furnace with the crucible and top poured into the moulds.
Table 1. Dimensions of cylindrical moulds
|
S/no. |
Mould outer Surface Diameter (mm) |
Diameter of Mould Cavity (mm) |
Thickness of Mould (mm) |
Depth of Mould Cavity (mm) |
|
1 2 3 |
125 175 200 |
100 100 100 |
12.5 37.5 50.0 |
100 100 100 |
Four thermocouples of K-type (Chrome/Alumel) were connected to a digital Paperless Temperature Recorder RD8900 with a temperature range of -200°C to 1370°C. The thermocouples were inserted at specified locations in the moulds with the centre of cast as reference position as shown in experimental setup of Figure 2.
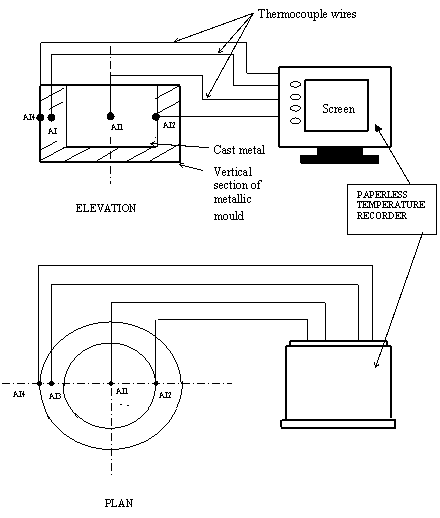
Figure 2. Schematic diagram of experimental setup for cylindrical mould
AI1 - Centre of cast (at radius, r = 0 for all moulds)
AI2 - Metal - Mould interface (at radius, r =50mm for all moulds)
AI3 - Half mould thickness (at r = 56.25 for 12.5mm thick mould; r = 68.75mm for 37.5mm thick mould, and r = 75.0mm for 50.0mm thick mould)
AI4 - Mould - Air interface (at r = 62.5mm for 12.5mm thick mould; r = 87.5mm for 37.5mm thick mould, and r = 100.0mm for 50.0mm thick mould)
The temperature readings from the Temperature Recorder were recorded at interval of 5 seconds in order to monitor the solidification and subsequent cooling process.
Results and discussion
Figures 3, 4 and 5 presents results of mould heating curves as well as the cast solidification and cooling curves for 12.5 mm, 37.5 mm and 50.0 mm thick mould.
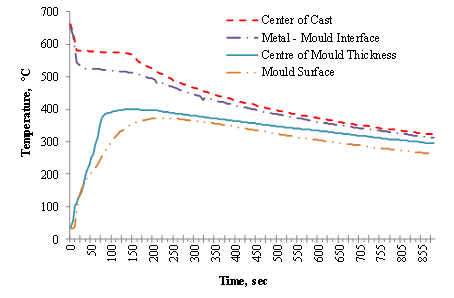
Figure 3. Variation of temperature with time of casting in 12.5 mm thick cylindrical mould
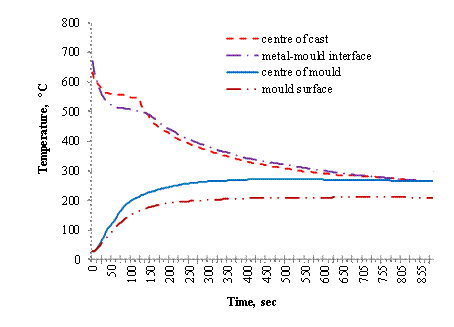
Figure 4. Variation of temperature with time of casting in 37.5 mm thick cylindrical mould
For Figures 3, 4 and 5 it is observed that at the metal/mould interface (AI2), the temperature decreases rapidly for several degrees and thereafter becomes relatively slow. The sudden change in the rate of decrease in temperature indicates faster cooling at the interface.
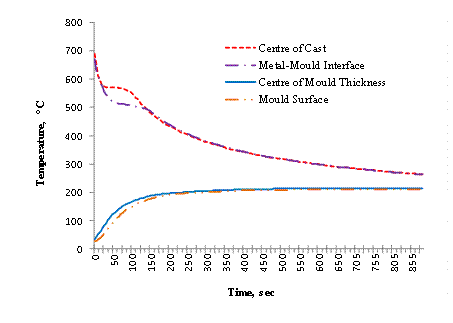
Figure 5. Variation of temperature with time of casting in 50 mm cylindrical mould
This leads to undercooling which is ascribed to the sudden release of latent heat of fusion at the mould wall. In the experiment performed, the interface thermocouple (AI2) could not show any measurable arrest of the undercooling, because solidification was completed at the interface almost instantaneously.
The Figures also show the mould heating curves obtained at the mould- air interface (AI4) and half mould thickness (AI3). Temperature rises more rapidly and higher in the 12.5 mm thick mould at the start of solidification until peak values of 339°C and 372°C are reached respectively before declining. A progressive rise in temperature is observed for the thicker moulds of 37.5 mm and 50.0 mm, with the 37.5 mm rising at a higher rate than the 50.0 mm thick mould.
Table 2. Solidification time for various thicknesses of cylindrical moulds
|
Mould Thickness (mm) |
Liquidus Temp (ºC) |
Solidus Temp (ºC) |
Solidification Time (sec) |
|
12.5 |
645 |
570 |
145 |
|
37.5 |
645 |
570 |
65 |
|
50.0 |
645 |
570 |
55 |
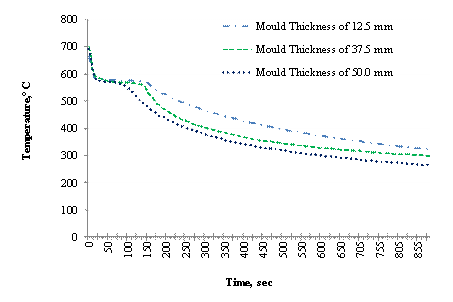
Figure 6. Variation of temperature with time at the centre of cast in cylindrical moulds
In order to determine effect of the various mould thicknesses on the rate solidification, the cooling curves at the center of the cast (AI1) for the three moulds thicknesses were compared as shown in Figure 6 from which the solidification time of the cast was estimated as presented in Table 2. The Table shows that solidification takes place much rapidly in the thicker moulds of 37.5 mm and 50.0 mm compared to the 12.5 mm mould. This is an indication that increase in mould thickness increases the rate of solidification. This is because increase in thickness increases the amount of heat absorbed by mould.
Conclusion
The experimental results have shown that mould thickness has influence on the solidification time of aluminium casting. Solidification time is longer for cylindrical mould with smaller mould thickness. As the thickness of mould increases solidification takes place at faster rate. This is due to the chilling effect of the mould which is also dependent on the heat content of the molten metal.
References
1. Gopinath V., Balanarasimman N., Effect of solidification parameters on the feeding efficiency of LM6 alluminium alloy casting, IOSR Journal of Mechanical and Civil Engineering, 2012, 4(2), p. 32-38.
2. Ozioko F. U., Casting of motorcycle piston from aluminium scrap using metallic mould, Leonardo Journal of Practices and Technologies, 2012, 11(21), p.82-92.
3. Ravi S., Josh D., Chougule R., Survey of computer applications in Indian foundry industry: benefits and concerns, 53rd Indian Foundry Congress, Institute of Indian Foundrymen, Kolkata, 2005.
4. Sheidi H. M., Investigation on properties of local zircon sand for sound casting: Azara-Lafia Deposit Nigeria, Research Journal in Engineering and Applied Sciences, 2012, 1(6), p. 404-407.
5. Adegbuyi P. A. O., Adedeji K. A., Raji N. A., Akinbuja A. O., The effect of cooling rate on the microstructure and mechanical properties of Al-Si alloy, Elixir Internal Journal of Mechanical Engineering, 2012, 52, p.11638-11644.
6. Madhusudhan, Narendranaath S., Kumar G. C. M., Mukunda P. G., Experimental study on rate of solidification of centrifugal casting, International Journal of Mechanical and Materials Engineering, 2010, 5(1), p. 101-105.
7. Madhusudhan, Narendranaath S., Mohankumar G. C., Mukunda P. G., Effect of mould wall on rate of solidification of centrifugal casting, International Journal of Engineering Science and Technology, 2010, 2(11), p. 6092-6096.
8. Madhusudhan, Narendxranath S., Mohankumar G. C., Effect of Mould wall temperature on rate of solidification of centrifugal casting, International Journal of Advanced Materials Science, 2013, 4(1), p. 37-42.
9. Bala K. C., Khan R. H., Abolarin M. S., Abubakre O. K., Investigation on the rate of solidification and mould heating in the casting of commercially pure aluminium in permanent moulds of varying thickness, IOSR Journal of Mechanical and Civil Engineering, 2013, 6(1), p. 33-37.
10. Rihan Y., Abd El-Bary, Numerical study on the effect of solidification parameters during the continuous casting of Al-Sn alloys, Online Journal of Mathematics and Statistics, 2010, 1(1), p. 8-12.
11. Bala K. C., Khan R. H., Experimental determination of the effect of mould thickness on the solidification time of aluminium alloy (Al-Mn-Ni-Si) casting in rectangular metallic moulds, International Journal of Engineering Research & Technology, 2013, 2(3), p.482-487.
12. Eman J. A, The influence of different casting method on solidification time and mechanical properties of Al –Sn castings, International Journal of Engineering Research & Technology, 2011, 11(6), p. 34-44.
13. Holman J. P., Heat transfer. McGraw Hill Inc, 8th edition, 1999.
14. Incropeara F. P., Dewitt D. P., Fundamentals of heat and mass transfer, John Wiley & Sons, 3rd edition, 1990.
15. Eduardo N. S., Noé C., Carlos A. S., Amauri G. Factors affecting solidification thermal variables along the cross-section of horizontal cylindrical ingots, Materials Science and Engineering , Elsevier, 2005, A(397), p. 239 - 248.
16. Rahul S., Modelling of heat transfer in solidification process, A term- report presented in mechanical engineering department, Indian Institute of Technology, Guwahati, 2005.