Gravimetric and electrochemical studies of corrosion inhibition potential of acid and ethanol extract of siam weed on mild steel
Sunday J. OLUSEGUN 1*, Elvis A. OKORONKWO 2, Anita E. OKOTETE 2, Olabisi A. AJAYI 1
1Corrosion and Electrochemical Laboratory, Department of Metallurgical and Materials Engineering, Federal University of Technology PMB 704, Akure, Ondo State, Nigeria
2Department of Chemistry, Federal University of Technology, PMB 704, Akure, Ondo State, Nigeria
* Corresponding author: arikawedy@yahoo.com, sjolusegun@futa.edu.ng
Abstract
The inhibition potency of acid and ethanol extract of siam weed was studied on the corrosion of mild steel in 1 M HCl using weight loss techniques, Tafel extrapolation, scanning electron microscopy and energy dispersive microscopy (SEM-EDS). The results from the weight loss revealed that the inhibition efficiency is dependent on the concentration of the extract and the temperature of the system. However, the ethanol extract has the highest inhibition efficiency of 83 %. The data obtained were found to be fitted into the adsorption models proposed by Langmuir and Freundlich. Morphological analysis indicates that the mild steel was well protected in the presence of the extracts. Tafel extrapolation shows that the extract acts as a mixed-type inhibitor.
Keywords
Mild steel; Corrosion inhibition; Tafel extrapolation; Siam weed; Adsorption isotherm; scanning electron microscopy and energy dispersive microscopy (SEM-EDS)
Introduction
Aesthetical values and mechanical strength of metallic materials are always compromised during their interaction with different environments such as acid and alkaline media. This leads to material degradation and has resulted to shutdown of many industrial plants, power plants, refineries and cumulatively caused excessive economic loss [1, 2]. In addition, corrosion products formed on degrading material surfaces have become deadly contaminants in pharmaceutical, dye and packaged goods industries and at the long run jeopardize the health of the consumers of these product [3]. However, due to the life-threatening consequences that corrosion brings, the protection of metallic materials has attracted the attention of corrosion scientists.
Among several methods that have been utilized to prevent metallic corrosion during acid pickling, descaling and oil well acidization is the addition of inhibitors. Corrosion inhibitors are chemicals substances which are added in small concentration to an environment and retard the occurrence of corrosion reactions [4]. It has been established that corrosion inhibitors protect metals by adsorbing on their surfaces which leads to the blockage of the reaction sites for metal dissolution to occur [5].
Inhibitors sourced from plants (Green corrosion inhibitors) have been proven to be environmentally friendly because they are biodegradable and do not contain heavy metals or other toxic compounds that could impinge the ecosystem [6]. Several researchers have reported the utilization of extracts of plants as inhibitors for the protection of metallic materials in different corrosive medium; Chenopodium Ambrosioides [7], Ficus asperifolia [8], Tithonia Diversifolia [9], Jatropha Curcas [10]. Other plants whose extract have been used as inhibitors are Nicotiana tabacum [11], Hunteria umbellata [12], cucumber [13], Brown Onion Peel [14], Geissopermum leave [15], Bamboo [16], Pectin (from citrus) [17], Carica papaya [18], Water Hyacinth [19]. Recently the exploration of animal wastes as corrosion inhibitors was reported [20, 21].
Siam weed (Chromolaena odorata) is an aggressive pioneer shrub species, and regarded as a very serious threat to agriculture in most invaded countries [22]. Chromolaena odorata has also been found to be a highly potent medicinal herb according to the traditional medicinal systems [23]. It has been reportedly used in livestock nutrition. The chemical composition of Chromolaena odorata consists of a diverse range of secondary chemicals, including flavonoids and terpenoids [24]. The presence of these phytochemical constituents makes C. odorata suitable for study of its corrosion inhibitory potentials.
Although, Obot et al. [25] had studied the inhibitory effect of ethanol extract of C. odorata leaves on mild steel, the study focused on H2SO4 solutions. According to Saratha and Vasudha [26], HCl is the most frequently used acid in industry for pickling, cleaning and de-scaling. Fernando et al. [4] added that hydrochloric acid solution is generally being used in the acidification operations in petroleum production. It becomes interesting to investigate the interaction of C. odorata extracts and mild steel in the HCl solution. The present study therefore seeks to investigate the corrosion inhibition potential of both acid and ethanol extracts of C. odorata leaves on the corrosion of mild steel in 1M HCl.
Material and method
Materials preparation
Mild steel of composition (wt%) as C (0.215), Si (0.258), Mn (0.467), S (0.009), P (0.007), Cr (0.021), Ni (0.001), Cu (0.017) and the rest Fe, was used for the study. The steel was mechanically cut to coupons of dimension, 1.8×1.4×0.4 cm. All reagents used for the study were analar grade and distilled water was used for the preparation.
Extraction of plant
The leaves of the plant were gathered within Akure, Nigeria, dried and pulverized. Acid extraction was done by soaking 10g of the pulverized leaves in a beaker containing 100 ml of 1M HCl, placed inside a water bath at 90ºC for 3 hours. It was cooled to room temperature and filtered. The filtrate was kept as the stock solution. Working solutions of different concentrations ranging from 1 to 5% v/v were prepared from the stock solution. Ethanol extraction was carried out by soaking 10 g of the pulverized leaves in ethanol and allowed to stay for 72 hours at room temperature, and then filtered. The filtrate was subjected to evaporation to leave the residue free of ethanol. The extract (residue) obtained was used to prepare working solutions of different concentrations ranging from 0.1 to 0.5 g/L.
Phytochemical analysis
Phytochemical analysis of the extract was carried out according to the method of Charu et al. [27]. A few drops of 1% NH3 solution was added to the aqueous extract in a test tube. A yellow colouration was observed to indicate that flavonoid is present. To determine tannins, 0.5 g of the extract was boiled in 20 ml of distilled water in a test tube and then filtered. 0.1 % FeCl3 was added to the filtrate and observed for brownish green or a blue black colouration, which shows the presence of tannins. Ammonia solution and conc. H2SO4 were used for the identification of flavonoid.
Gravimetric test
The polished and pre-weighed mild steel coupons were immersed in 50ml of 1 M HCl solution in the absence and presence of the respective Chromolaena odorata leaves extracts and maintained at 303, 313, 323, and 333K in a water bath for 4 hours. After which the coupons were removed, rinsed in distilled water, ethanol and then weighed. The results obtained were used to evaluate corrosion rate, inhibition efficiency, adsorption isotherms and thermodynamics parameters.
The inhibition efficiency (IE %) was calculated using the relationship:
IE% = (1- CRp/CRa) × 100 (1)
where CRp and CRa correspond to the corrosion rates in the presence and absence of inhibitor respectively.
Corrosion rate was calculated using the equation:
CR (mgh-1cm-2) = ∆W/AT (2)
where CR, ∆W, A and T are corrosion rate, weight loss, area of the coupon and time respectively.
The data obtained from the weight loss were fitted into Langmuir and Freundlich adsorption isotherm by using equation 3 and 4 respectively.
![]() (3)
(3)
where C is the inhibitor concentration, Ө is the surface coverage and kads is the adsorption equilibrium constant.
logΘ=logKf +logC/n (4)
where Kf is Freundlich isotherm constant, n is adsorption intensity.
Tafel extrapolation
The electrochemical experiments were carried out using VERSTAT 4 instrument. The experiments were performed using a three-electrode corrosion cell set-up comprising of mild steel as the working electrode (surface area = 1cm2), saturated silver/silver chloride as reference electrode, and platinum rod as counter electrode. The test electrolyte was 1M solution of HCl in the presence and absence of the extract.
Potentiodynamic polarization measurements were carried out using scan rate of 1.0 mV/s at potential initiated at -250 mV to + 250 mV with respect to OCP. After each experiment, the electrolyte and the test sample were replaced. The linear Tafel segments of the anodic and cathodic curves were extrapolated to corrosion potential to obtain the corrosion current density (Icorr) and corrosion potential (Ecorr). Anodic Tafel slope (βa) and cathodic Tafel slopes (βc) were determined from the experimental curve. The IEIcorr (% ) was calculated using the current densities.
IEIcorr(%) = (Iºcorr - Icorr)/ Iºcorr × 100 (5)
where IEcorr, Iºcorr and Icorr are the inhibition efficiency, current density in the absence and presence of inhibitor respectively.
Scanning electron microscopy
The surface morphology of the mild steel before and after immersion was examined using JSM 7600F Jeol ultra-high resolution field emission gun scanning electron microscope equipped with energy dispersive spectroscopy to analyze the elements on the surface.
Results and discussion
Effect of extract concentration
The plots of weight loss against exposure time for the corrosion inhibition of mild steel in the presence and absence of Ethanol Extract of Chromolaena Odorata (EECO) and Acid Extract of Chromolaena Odorata (AECO) are shown in Figure 1 and 2.
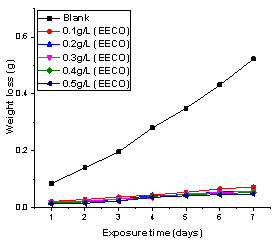
Figure 1. Variation of weight loss with time for the corrosion of mild steel in 1M HCl containing various concentrations of EECO
From the plots, the weight loss increased with increased in immersion time but decreased with increased in inhibitor concentration. The decreased in weight loss in the presence of inhibitor was due to the adsorption of the phytochemical constituents in the extract on the surface of mild steel cuopon.
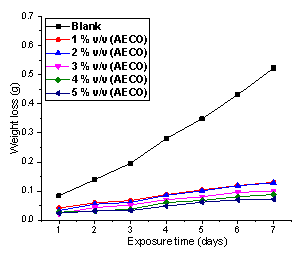
Figure 2. Variation of weight loss with time for the corrosion of mild steel in 1M hydrochloric acid containing various concentrations of AECO
According to Peter et al. [28], these compounds can absorb on the metal surface via the lone pairs of electrons present on their oxygen, sulphur and nitrogen atoms. Saratha and Vasudha [26] added that the adsorption of such compounds on the metal surface create a barrier for charge and mass transfer thereby minimizing corrosion reaction.
Comparing Figures 1 and 2, after the 7th day, the addition of 0.5 g/l of EECO resulted in decreasing the weight loss from 0.5227 g in the absence of the extract to 0.0475 g. However, the addition of 5%v/v of AECO retarded the weight loss to 0.0723 g. Similar trend was also reported by Alaneme et al. [29]. These observations suggest that Chromolaena odorata is a potential inhibitor for the corrosion of mild steel in hydrochloric acid.
The phytochemical results showed that EECO contains 119 mg/g saponin, 0.265 mg/g tannin and 7.562 mg/g flavonoid, whereas, AECO contains 32.9, 0.165 and 4.409 mg/g of saponin, tannin and flavonoid respectively. From these results EECO contains more of the phytochemical constituents than AECO this would have been responsible for better protection that EECO offered compared to AECO.
Effect of temperature on inhibition efficiency
The plots of inhibition efficiency against temperature are shown in Figure 3 and 4. From these Figures, inhibition efficiency decreases with increased in temperature for all concentrations of the plant extracts. Decrease in inhibition efficiency with increase in temperature is due to desorption of some of the adsorbed inhibitor molecules from the metal surface at higher temperatures. This indicates that the extracts were physically adsorbed on the metal surface. For a physical adsorption mechanism, inhibition efficiency decreases with temperature while for a chemical adsoption mechanism, values of inhibition efficiency increase with temperature [30].
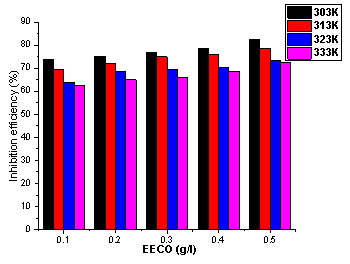
Figure 3. Variation of inhibition efficiency of EECO with concentration at 303-333K
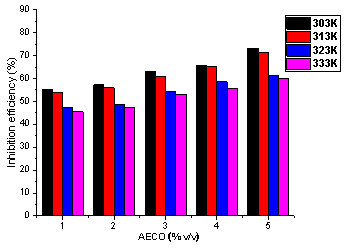
Figure 4. Variation of inhibition efficiency of AECO with concentration at 303-333K
According to Alinor and Ejimeke [31] at higher temperature, the average kinetic energy of components of extracts increases, thus making adsorption between components of extracts and metal surface insufficient to retain the species at the binding sites. This could lead to desorption or cause species to bounce off surface of the metal instead of colliding and combining with it. Therefore, increase in temperature is associated with decrease in stability of adsorbed extract components on the metal surface [29, 32]. Thus, the inhibition of mild steel using C. odorata will be best applicable at lower temperature. It was observed that highest percentage inhibition efficiency [EECO (83.2%) and AECO (73.34 %)] was attained at the highest concentration of the extract.
Adsorption isotherms
Adsorption isotherms are very important in determining the mechanism of corrosion reactions [33, 34]. The plot of C/Θ against C gives a linear plot as shown in Figure 5 and 6 indicating that the adsorption of the inhibitor molecules on the surface of mild steel is consistent with Langmuir isotherm and the slopes obtained are unity.
The values of correlation coefficient (R2) and adsorption coefficient (Kads) are shown in Tables 1 and 2. The values of Kads show that adsorption coefficient decreased with increase in temperature indicating that inhibition efficiency decreases with temperature. Kads denotes the strength between adsorbate and adsorbent [35]. The higher value of Kads at 303 K implies that more of the extracts were adsorbed onto the surface of mild steel leading to a greater surface coverage.
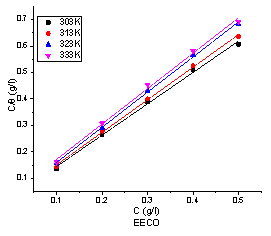
Figure 5. Langmuir adsorption isotherm plot for the adsorption of EECO on the surface of mild steel in 1 M HCl
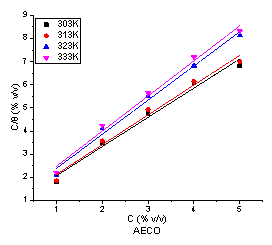
Figure 6. Langmuir adsorption isotherm plot for the adsorption of AECO on the surface of mild steel in 1 M HCl
The plot of logΘ against log C is shown in Figure 7 and 8. The constant Kf is an approximate indicator of adsorption capacity, while 1/n is a function of the strength of adsorption in the adsorption process [36]. If 1/n = 1 then the partition between the two phases are independent of the concentration, if it is <1, a normal adsorption and if > 1, then there is cooperative adsorption [34].
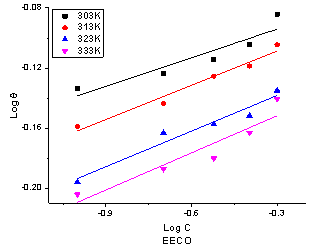
Figure 7. Freundlich adsorption isotherm plot for the adsorption of EECO on the surface of mild steel in 1 M HCl
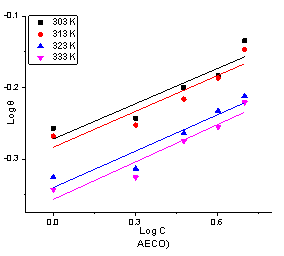
Figure 8. Freundlich adsorption isotherm plot for the adsorption of AECO on the surface of mild steel in 1 M HCl
The value of 1/n for all the extract as shown in Tables 1 and 2 are less than 1 indicating that adsorption of the phytochemical constituents on the surface on the mild steel is a normal adsorption process and is dependent on the concentration of the extract.
Table 1. Langmuir and Freundlich isotherm parameter for EECO
|
Models |
Parameter |
303K |
313K |
323K |
333K |
|
Langmuir |
Kads |
38.22 |
36.15 |
35.95 |
26.27 |
|
R2 |
0.9986 |
0.9994 |
0.9994 |
0.9980 |
|
|
Freundlich |
KF |
0.841 |
0.820 |
0.768 |
0.746 |
|
1/nF |
0.0636 |
0.0762 |
0.0791 |
0.0825 |
|
|
R2 |
0.9274 |
0.9853 |
0.9762 |
0.9402 |
The values of Kf as shown in Table 1 and 2 decrease as the temperature increases indicating that plant constituents are physically adsorbed on the surface of mild steel this conforms to the results obtained from Langmuir isotherm model.
Table 2. Langmuir and Freundlich isotherm parameter for AECO
|
Models |
Parameter |
303K |
313K |
323K |
333K |
|
Langmuir |
Kads |
1.24 |
1.19 |
1.10 |
1.03 |
|
R2 |
0.9915 |
0.9924 |
0.9957 |
0.9949 |
|
|
Freundlich |
KF |
0.534 |
0.520 |
0.457 |
0.440 |
|
1/nF |
0.1642 |
0.1671 |
0.1696 |
0.1742 |
|
|
R2 |
0.9301 |
0.9368 |
0.9500 |
0.9560 |
Tafel extrapolation study
Tafel polarization plots obtained for mild steel in the absence and presence of the extract in 1 M HCl at room temperature is presented in Figure 9 and 10. Corrosion potential (Ecorr), corrosion current density (Icorr), anodic Tafel constant (βa), cathodic Tafel constant (βc), and the corresponding inhibition efficiencies are listed in Table 3 and 4. It is obvious from the Figures that the plant extracts reduced both the anodic and cathodic current significantly. The addition of the extract reduced the corrosion current densities from 480.1 μA/cm2 to 38 μA/cm2 with the addition of 0.5g/l of EECO extract; and to162.5 μA/cm2 with addition of 5% v/v AECO extract.
However, there is no significant difference in the corrosion potential (Ecorr) of the blank and the inhibited samples. A corrosion inhibitor has been termed anodic or cathodic type when the deviation in the Ecorr value is more than 85 mV [37]. As shown in Tables 3 and 4, the deviation in the Ecorr obtained in the presence and absence of the extract is less than 20mV this confirms that the extracts act as mixed-type inhibitor.
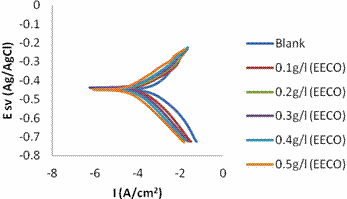
Figure 9. Tafel polarization curves for the mild steel in the absence and presence of EECO
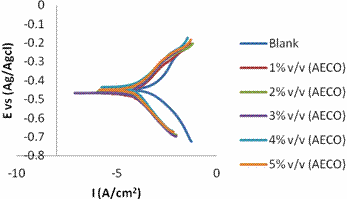
Figure 10. Tafel polarization curves for the mild steel in the absence and presence of AECO
Table 3. Tafel polarization parameters and corresponding inhibition efficiency for the corrosion of mild steel in 1M HCL containing different concentrations of EECO
|
EECO( g/l) |
Ecorr (mv/SCE) |
Icorr (μA/cm2) |
βa (mV/decade) |
βc (mV/decade) |
IEIcor (%) |
|
Blank |
-449 |
480.1 |
103.7 |
96.8 |
- |
|
0.1 |
-444 |
131.4 |
68 |
87 |
72.6 |
|
0.2 |
-440 |
86.9 |
63 |
97 |
81.9 |
|
0.3 |
-437 |
78.8 |
66.7 |
93.8 |
83.5 |
|
0.4 |
-446 |
74 |
84 |
100.6 |
84.6 |
|
0.5 |
-448 |
38 |
82 |
94.4 |
92.1 |
Table 4. Tafel polarization parameters and corresponding inhibition efficiency for the corrosion of mild steel in 1M HCL containing different concentrations of AECO
|
EECO (%) v/v |
Ecorr (mv/SCE) |
Icorr (μA/cm2) |
βa (mV/decade) |
βc (mV/decade) |
IEIcor(%) |
|
Blank |
-449 |
480.1 |
103.7 |
96.8 |
- |
|
1 |
-436 |
198.6 |
90 |
113 |
58.6 |
|
2 |
-456 |
171 |
95 |
129 |
64.38 |
|
3 |
-464 |
171.1 |
97 |
122 |
64.38 |
|
4 |
-458 |
168.5 |
105 |
143 |
64.9 |
|
5 |
-467 |
162.5 |
118 |
122 |
66.2 |
Corrosion current density (Icorr), corrosion potential (Ecorr), anodic Tafel slope (βa), cathodic Tafel slopes (βc) and percentage inhibition efficiency IEIcorr (% ).
Surface morphology analysis
SEM micrographs of the mild steel before immersion, after immersion in hydrochloric acid with and without inhibitors are presented in Pictures 1-4.
Surface analysis using scanning electron microscopy provided more information on the inhibition mechanism of the extract on the surface of mild steel. The surface of the mild steel in the absence of inhibitor revealed severe damage by the acid. Whereas, the addition of the extract to the test solution, protected the metal this led to less surface deterioration.
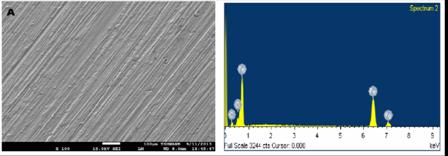
Picture 1. SEM micrograph and EDS spectra of plain mild steel

Picture 2. SEM micrograph and EDS spectra of mild steel immersed in hydrochloric acid without inhibitor

Picture 3. SEM micrograph and EDS spectra of mild steel immersed in hydrochloric acid in the presence of EECO

Picture 4. SEM micrograph and EDS spectra of mild steel immersed in hydrochloric acid in the presence of AECO
The EDS (Energy dispersive spectroscopy) spectra showed reduction in Fe percentage of the blank compared to the inhibited mild steel. From the EDS analysis the % weight of Fe before immersion was 90.79, mean while after immersion in the absence and presence of EECO and AECO are 51.54, 85.12 and 76.63 respectively. Alaneme et al., [38] also reported similar observation. This support the claim that the phytochemical constituents adsorbed on the surface of the mild steel prevented its severe dissolution.
Conclusions
The results obtained in the study have shown that extract of Chromolaena odorata leaves is potential inhibitor for mild steel in hydrochloric acid that is often use in oil well acidization. The inhibition efficiency increases with the increase in the concentration of the extract but decreases as the temperature increases. The decreased in the inhibition efficiency with increased in the temperature confirms a physical adsorption mechanism. The adsorption mechanism of the extract on the surface of mild steel conforms to the Langmuir and Freundlich adsorption models. The result of the SEM-EDS of the mild steel showed that the inhibition was due to the adsorption of the extract molecules on the metal surface. Tafel polarization results showed that the extract is a mixed-type inhibitor.
References
1. Amabogha B., Corrosion in Thermal Energy Generating Plants, International Journal of Engineering and Applied Sciences, 2013, 4, p. 29-35.
2. Fontana M. G., Corrosion Engineering, New York: McGraw-Hill Book Company, 3rd Ed., 1986.
3. Satapathy A. K., Gunasekaran G., Sahoo S. C., Kumar A., Rodrigues P. V., Corrosion inhibition by Justicia gendarussa plant extract in hydrochloric acid solution, Corrosion Sci., 2009, 51, p. 2848-2856.
4. Fernando B. M., Thais M. B., Utsch M., Antonio C. M. R., Performance of corrosion inhibitors based on n-butylamine in protection of carbon steel in hydrochloric acid solution, International Refereed Journal of Engineering and Science, 2014, 3, p. 38-42.
5. De Sousa F. S., Spinelli A., Caffeic acid as a green corrosion inhibitor for mild steel, Corrosion Sci., 2009, 51, p. 642-649.
6. Sethuraman M. G., Aishwarya V., Kama C., Edison T. J. I., Studies on ervatinine - the anticorrosive phytoconstituent of Ervatamia coronaria, Arab. J. Chem., 2012, doi : 10.1016/j.arabjc.2012.10.013
7. Bammou L., Belkhaouda M., Salghi R., Benali O., Zarrouk A., Zarrok, H., Hammouti B., Corrosion inhibition of steel in sulfuric acidic solution by the Chenopodium Ambrosioides Extracts, Journal of the Association of Arab Universities for Basic and Applied Sciences, 2014, 16, p. 83-90.
8. Fadare O. O., Okoronkwo A. E., Olasehinde E. F., Assessment of anti-corrosion potentials of extract of Ficus asperifolia -Miq (Moraceae) on mild steel in acidic medium, African Journal of Pure and Applied Chemistry, 2016, 10, p. 8-22.
9. Alaneme K. K., Olusegun S. J., Corrosion Inhibition Performance of Lignin Extract of Sun Flower (Tithonia Diversifolia) on Medium Carbon Low Alloy Steel Immersed in H2SO4 Solution, Leonardo Journal of Science, 2012, 20, p. 59-70.
10. Olusegun S. J., Adeiza B. A., Ikeke K. I., Bodunrin M. O., Jatropha Curcas Leaves extract as Corrosion Inhibitor for Mild Steel in 1M Hydrochloric Acid, Journal of Emerging Trends in Engineering and Applied Sciences, 2013, 4, p. 138-143.
11. Olasehinde E. F., Olusegun S. J., Adesina A. S., Omogbehin S. A., Momoh-Yahayah, H., Inhibitory Action of Nicotiana tabacum Extracts on the Corrosion of Mild Steel in HCl: Adsorption and Thermodynamics Study, Nat Sci., 2013, 11, p. 83-90.
12. Alaneme K. K., Olusegun S. J., Adelowo O. T., Corrosion inhibition and adsorption mechanism studies of Hunteria umbellata seed husk extracts on mild steel immersed in acidic solutions, Alexandria Eng. J., 2016, 55, p. 673-681.
13. Ghadah M., Al-Senani, Corrosion Inhibition of Carbon Steel in acidic chloride medium by Cucumis Sativus (cucumber) Peel Extract, Int. J. Electrochem. Sci., 2016, 11, p. 291-302.
14. Ferreira K. C. R., Cordeiro R. F. B., Nunes J. C., Orofino H., Magalhães M., Torres A. G., D'Elia E., Corrosion Inhibition of Carbon Steel in HCl Solution by Aqueous Brown Onion Peel Extract, Int. J. Electrochem. Sci., 2016, 11, p. 406-418
15. Faustin M., Maciuk A., Salvin P., Roos C., Lebrini M., Corrosion inhibition of C38 steel by alkaloids extract of Geissopermum leave in 1 M Hydrochloric acid: Electrochemical and phytochemical studies, Corrosion Science, 2015, 92, p. 287-300.
16. Xianghong L., Shuduan D., Xiaoguang X., Hui F., Inhibition effect of bamboo leaves extract on steel and zinc in citric acid solution, Corrosion Sci., 2014, 87, p.15-26.
17. María V., Fiori-Bimbi, Patricia E., Alvarez V., Claudio Gervasi A., Corrosion inhibition of mild steel in HCl solution by pectin, Corrosion Sci., 2015, 92, p. 192-199.
18. Ajanaku K. O., Aladesuyi O., Anake W. U., Edobor-Osoh A., Ajanaku C. O., Siyanbola T. O., Akinsiku A. A., Inhibitive properties of Carica papaya leaf extract on Aluminium in 1.85M HCl, Journal of Advances in Chemistry, 2014, 8, p. 1651-1659.
19. Oloruntoba D. T., Abbas J. A., Olusegun S. J., Water Hyacinth (eichhornia crassipes) Leaves Extract as Corrosion Inhibitor for AISI 1030 Steel in Sea Water, Proceedings of 4th West Africa Built Environment Research (WABER), Abuja, Nigeria, 2012, p. 1131-1140.
20. Okoronkwo A. E., Olusegun S. J., Oluwasina O. O., The inhibitive action of chitosan extracted from Archachatina marginata shells on the corrosion of plain carbon steel in acid media, Anti-Corrosion Methods and Materials, 2015, 62, p. 13-18.
21. Olusegun S. J., Oluwasina O. O., Alaneme K. K., Olubambi P. A., Corrosion inhibition of mild steel in acidic solution by cow dung extract as an eco-friendly inhibitor, J. Mater. Environ. Sci., 2016, 7, p. 1086-1097.
22. Muniappan R., Bamba J., Biological Control of Chromolaena odorata: Successes and Failures. Proceedings of the X International Symposium on Biological Control of Weeds. Montana State University, Bozeman, Montana, USA Neal R. Spencer [ed.]., 2000, p. 81-85.
23. Ayyanar M., Ignacimuthu S., Herbal medicines for wound healing among tribal people in Southern India: Ethnobotanical and Scientific evidences, International Journal of Applied Research in Natural Products, 2009, 2, p. 29-42.
24. Aro S. O., Osho I. B., Aletor V. A., Tewe O. O., Review on Chromolaena odorata in livestock nutrition, Journal of Medicinal Plants Research, 2009, 3, p. 1253-1257.
25. Obot I. B., Ebenso E. E., Zuhair M. G., Eco-friendly Corrosion Inhibitors: Adsorption and Inhibitive Action of Ethanol Extracts of Chlomolaena Odorata L. for the Corrosion of Mild Steel in H2SO4 Solutions, Int. J. Electrochem. Sci., 2012, 7, p. 1997-2008.
26. Saratha R., Vasudha V. G., Emblica Officinalis (Indian Gooseberry) Leaves Extract as Corrosion Inhibitor for Mild Steel in 1N HCl Medium, E-Journal of Chemistry, 2010, 7, p. 677-684.
27. Charu A. C., Sanjeev M., Heena D., Phytochemical Screening and Evaluation of Biological Activities of Some Medicinal Plants of Phagwara, Punjab, Asian Journal of Chemistry, 2012, 24, p. 5903-5905.
28. Peter C. O., Eno E. E., Udofot J. E., Azadirachta Indica Extracts as Corrosion Inhibitor for Mild Steel in Acid Medium, Int. J. Electrochem. Sci., 2010, 5, p. 978-993.
29. Alaneme K. K., Daramola Y. S., Olusegun S. J., Afolabi A. S., Corrosion Inhibition and Adsorption Characteristics of Rice Husk Extracts on Mild Steel Immersed in 1M H2SO4 and HCl Solutions, Int. J. Electrochem. Sci., 2015, 10, p. 3553-3567.
30. Ebenso E. E., Eddy N. O., Odiongenyi A. O., Corrosion inhibitive properties and adsorption behaviour of ethanol extract of Piper guinensis as a green corrosion inhibitor for mild steel in H2SO4, African Journal of Pure and Applied Chemistry, 2008, 2, p. 107-115.
31. Alinnor I. J., Ejikeme P. M., Corrosion Inhibition of Aluminium in Acidic Medium by Different Extracts of Ocimum Gratissimum, American Chemical Science Journal, 2012, 2, p. 122-135.
32. Singh A., Ahamad I., Yadav D. K., Singh V. K., Quraishi M. A., The effect of environmentally benign fruit extract of shahjan (Moringa oleifera) on the corrosion of mild steel in hydrochloricacid solution, Chem. Eng. Commun., 2012, 199(1), p. 63-77.
33. Ambrish Singh, Vinod Kumar Singh, Quraishi MA, Inhibition of Mild Steel Corrosion in HCl Solution Using Pipali (Piper longum) Fruit Extract, Arabian Journal for Science and Engineering, 2013, 38, p. 85-97.
34. Dada A. O., Olalekan A. P., Olatunya A. M., Dada O., Langmuir, Freundlich, Temkin and Dubinin-Radushkevich Isotherms Studies of Equilibrium Sorption of Zn2+ unto Phosphoric Acid Modified Rice Husk, IOSR Journal of Applied Chemistry, 2012, 3, p. 38-45.
35. Ating E. I., Umoren S. A., Udousoro I. I., Ebenso E. E., Udoh A. P., Leaves extract of Ananas sativum as green corrosion inhibitor for aluminium in hydrochloric acid solutions, Green Chemistry Letters and Reviews, 2010, 3, p. 61-68.
36. Voudrias E., Fytianos F., Bozanim E., Sorption Description isotherms of Dyes from aqueous solutions and Waste Waters with Different Sorbent materials, Global Nest, The Int. J., 2002, 4, p. 75-83.
37. Deyab M. A., Adsorption and inhibition effect of Ascobyl palmitate on corrosion of carbon steel in ethanol blended gasoline containing water, Corrosion Sci., 2015, 80, p. 359-365.
38. Alaneme K. K., Olusegun S. J., Alo A. W., Corrosion inhibitory properties of elephant grass (Pennisetum purpureum) extract: Effect on mild steel corrosion in 1 M HCl solution, Alexandria Engineering Journal, 2016, 55, p. 1069-1076.